Advertisements
Advertisements
प्रश्न
No chemical reaction takes place when granules of a rusty-brown solid A are mixed with the powder of another solid B. However, when the mixture is heated, a reaction takes place between its components. One of the products C is a metal and settles down in the molten state while the other product D floats over it. It was observed that the reaction is highly exothermic.
(a) What could the solids A and B be?
(b) What are the products C and D most likely to be?
(c) Write the chemical equation for the reaction between A and B leading to the formation of C and D. Mention the physical states of all the reactants and products in this equation and indicate the heat change which takes place.
(d) What is the special name of such a reaction? State one use of such a reaction.
(e) Name any two types of chemical reactions under which the above reaction can be classified.
Advertisements
उत्तर
(a) The rusty-brown solid A is iron (III) oxide and another solid B is aluminium. Mixing of iron granules with aluminium powder will have no reaction.
(b) The product are likely to be C (iron) and D (aluminium oxide). Once the mixture of iron (III) oxide granules and aluminium powder is heated, aluminium acts as reducing agent. Aluminium reduces iron (III) oxide to iron and aluminium powder oxidises to aluminium oxide with the liberation of excess heat.
(c) 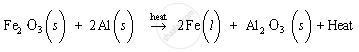
When A (iron (III) oxide) and B (aluminium powder) are heated, iron and aluminium oxide get produced with the liberation of excess heat. Thus, the product C is iron and product D is aluminium oxide. As excess heat is liberated in the reaction, the product C, i.e., iron metal, is in the molten state.
(d) The special name given to this reaction is thermite reaction. This reaction is widely used to weld broken hard substances like iron in railway tracks. This process of welding is called thermite welding.
(e) The above said reaction can be classified into displacement reaction and oxidation-reduction reaction. Aluminium, being more reactive than iron, displaces iron in iron (III) oxide, undergoing displacement reaction. Aluminium also acts as a reducing agent and reduces iron (III) oxide to iron metal and gets oxidised to aluminium oxide, undergoing oxidation-reduction reaction.
APPEARS IN
संबंधित प्रश्न
Tinning : Tin : : Galvanizing : _________
Write two methods of preventing the rusting of iron.
Why do we apply paint on iron articles?
Explain why Iron sheets are coated with zinc during galvanization.
What special name is given to the corrosion of iron?
Name the metal which is used for galvanising iron.
What is the corrosion of iron known as?
If copper is kept exposed to damp air for a considerable time, it gets a green coating on its surface. This is due to the formation of:
(a) hydrated copper sulphate
(b) copper oxide
(c) basic copper carbonate
(d) copper nitrate
Explain with reason:
Roasting is carried out on sulphide ores and not on carbonate ores.
Answer the following question:
What is corrosion? Do gold ornaments corrode? Justify.
What is ‘rusting’?
_______ is an alloy made from iron, carbon and chromium.
Find the odd one out and give its explanation.
Observe the following figure and write the answer of the question.

- Which process is shown in the figure?
- Explain the chemical reaction shown in the figure.
- Write the reactions on anode and cathode.
Give preventive methods by giving examples of corrosion?
Give the equation for the formation of rust.
Copper objects lose their shine and form green coating of ____________.
Gold plated ornaments is the example of ______.
Give scientific reasons.
Silver amalgam is used for filling dental cavities.
