Advertisements
Advertisements
प्रश्न
Is it always necessary to use red light to get a photoelectric effect?
Advertisements
उत्तर
No, the broad wavelength and low energy of red light, photons do not have sufficient energy to pull an electron out of its orbital.
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Polychromatic (containing many different frequencies) radiation is used in an experiment on the photoelectric effect. The stopping potential ______.
It is observed in an experiment on the photoelectric effect that an increase in the intensity of the incident radiation does not change the maximum kinetic energy of the electrons. Where does the extra energy of the incident radiation go? Is it lost? State your answer with explanatory reasoning.
As the intensity of incident light increases ______
Explain the concept of the photoelectric effect.
State Einstein photoelectric equation.
State Einstein’s photoelectric equation. Explain all characteristics of the photoelectric effect, on the basis of Einstein’s photoelectric equation.
With the help of a circuit diagram describe the experiment to study the characteristics of the photoelectric effect. Hence discuss any 2 characteristics of the photoelectric effect.
The energy of the incident photon on the metal surface is 3 W and then 5 W, where W is the work function for that metal. The ratio of velocities of emitted photoelectrons is ______.
The kinetic energy of the most energetic photoelectron emitted from a metal surface is doubled when the wavelength of the incident radiation is reduced from λ1 to λ2. The work function of the metal is ______
Which one of the following is TRUE in photoelectric emission?
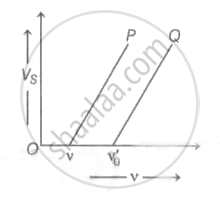
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
If the maximum kinetic energy of emitted electrons in photoelectric effect is 3.2 × 10-19 J and the work-function for metal is 6.63 × 10-19 J, then stopping potential and threshold wavelength respectively are
[Planck's constant, h = 6.63 × 1034 J-s]
[Velocity of light, c = 3 × 108 `"m"/"s"`]
[Charge on electron= 1.6 × 10-19 C]
Photoelectrons emitted from a metallic surface are initially ____________.
In photoelectric experiment, if both the intensity and frequency of the incident light are doubled, then the saturation of photoelectric current ______.
Light of wavelength `lambda` strikes a photo-sensitive surface and electrons are ejected with kinetic energy E. If the kinetic energy is to be increased to 2E, the wavelength must be changed to `lambda'` where ____________.
In photoelectric effect, for a light of different intensities but of same frequency, the stopping potential for a given metal is ____________.
Light of frequency 2 times the threshold frequency is incident on a photo sensitive material. If the frequency is made `1/3`rd and intensity is doubled then the photocurrent will ______.
When light of wavelength 'λ' is incident on a photosensitive surface, the stopping potential is 'V'. When light of wavelength '3λ' is incident on the same surface, the stopping potential is `"V"/6`. Threshold wavelength for the surface is _______.
When a surface 1 cm thick is illuminated by light of wavelength 'λ', the stopping potential is 'V0'. When the same surface is illuminated by light of wavelength '3λ', the stopping potential is `"V"_0/6`. The threshold wavelength for the metallic surface is ______.
The work function of a substance is 4.0 eV. The longest wavelength of light that can cause photo-emission from this substance is approximately (h = 6.63 × 10-34 Js)[1eV = 1.6 × 10-19 J]
Following graphs show the variation of stopping potential corresponding to the frequency of incident radiation (F) for a given metal. The correct variation is shown in graph (v0 = Threshold frequency).
When the work function of a metal increases, maximum kinetic energy of emitted photoelectrons ____________.
The stopping potential in the context of photoelectric effect depends on the following property of incident electromagnetic radiation ______.
The wavelength of light incident on a metal surface is reduced from 300 nm to 200 nm (both are less than threshold wavelength). What is the change in the stopping potential for photoelectrons emitted from the surface will be ______ V. (Take h = 6.6 × 10-34 J-s)
When ultraviolet light of wavelength 100 nm is incident upon a sample of silver metal, a potential difference of 7.7 volt is required to stop the photoelectrons from reaching the collector plate. The potential required to stop photo electrons when light of wavelength 200 nm is incident upon silver is ______.
The maximum kinetic energy of the photoelectrons ejected will be ______ eV when the light of wavelength 350 nm is incident on a cesium surface. The work function of cesium = 1.9 eV.
A point isotropic light source of power P = 12 watts is located on the axis of a circular mirror of radius R = 3 cm. If the distance of the source from the centre of the mirror is a = 39 cm and the reflection coefficient of the mirror is α = 0.70 then the force exerted by the light ray on the mirror is ______ × 10-10 N.
A charged dust particle of radius 5 × 10-7 m is located in a horizontal electric field having an intensity of 6.28 × 105 V/m. The surrounding medium is air with a coefficient of viscosity η = 1.6 × 10-5 N-s/m2. If the particle moves with a uniform horizontal speed of 0.02 m/s, the number of electrons on it is ______.
A parallel plate capacitor with air between the plates has capacitance 9 pF. The separation between the plates becomes thrice and the space between them is filled with a medium of dielectric constant 6. The capacitance becomes 'x' pF. The value of 'x' is ______.
