Advertisements
Advertisements
प्रश्न
Explain the reactions involved in the preparation of viscose rayon.
Advertisements
उत्तर
- Viscose rayon is a semisynthetic fibre which is regenerated cellulose.
- Cellulose in the form of wood pulp is transformed into viscose rayon. Cellulose is a linear polymer of glucose units and has the molecular formula [(C6H10O5)n]. A modified representation of the molecular formula of cellulose is Cell-OH.
- Method of preparation:
a. Cellulose in the form of wood pulp is treated with concentrated NaOH solution to get fluffy alkali cellulose.
b. It is then converted to xanthate by treating with carbon disulphide.
c. On mixing with dilute NaOH, it gives viscose solution which is extruded through spinnerets of the spinning machine into the acid bath when regenerated cellulose fibres precipitate. - Reactions involved in the preparation of viscose rayon:
\[\ce{\underset{\text{(Cellulose pulp wood)}}{Cell - OH + NaOH} -> \underset{\text{(Akali cellulose)}}{Cell - O- Na+} + H2O}\]
\[\begin{array}{cc}
\ce{Cell - O- Na+ + CS2 ->Cell - O - C - S- Na+}\\
\phantom{............................}||\\
\phantom{............................}\ce{\underset{\text{(Cellulose xanthate)}}{S}}
\end{array}\]
\[\begin{array}{cc}
\ce{Cell - O - C - S- Na+ + H2O -> Cell - O - C - SH + NaOH}\\
\phantom{..}||\phantom{..................................}||\phantom{.....}\\
\phantom{..}\ce{S}\phantom{..................................}\ce{S}\phantom{.....}
\end{array}\]
\[\begin{array}{cc}
\ce{Cell - O - C - SH + H2O -> \underset{\text{(Viscose rayon) (Regenerated cellulose filaments)}}{Cell - OH + CS2}}\\
||\phantom{.........................................}\\
\ce{S}\phantom{.........................................}
\end{array}\]
संबंधित प्रश्न
Based on molecular forces, what type of polymer is neoprene?
Write the formulae of the raw materials used for preparation of Dextran.
Write the structure of melamine.
Choose the correct option from the given alternatives.
Which of the following is made up of polyamides?
Answer the following in one sentence.
Identify 'B' in the following reaction:
\[\ce{H2N -(CH2)6 - NH2 + HOOC - (CH2)4 - COOH ->[N2][533 K]}\]'B'
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Answer the following.
Write the reaction of the formation of Terylene.
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Identify condensation polymers and addition polymers from the following.
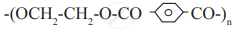
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Monomer used for preparation of polyacrylonitrile is _____________
Monomers ethylene glycol and terephthalic acid undergo condensation polymerization to give polymer called ___________
Monomer of natural rubber is __________
Write a chemical reaction for the preparation of the following polymer.
polyacrylonitrile
Write preparation of low density polythene.
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
Define rubber.
Which of the following is used as a substitute for wool?
The following structure represents the polymer:
\[\begin{array}{cc}
\ce{[-C-CH2-NH-C-(-CH2)5 NH -]_{{n}}}\\
\phantom{}||\phantom{.............}||\phantom{................}\\
\phantom{}\ce{O}\phantom{.............}\ce{O}\phantom{................}
\end{array}\]
How many isoprene units are present in abscisic acid?
Identify the INCORRECT match.
Which of the following is the monomer of neoprene?
Novolac is obtained from ____________.
Identify the polymer obtained by polymerization of n moles of acrylonitrile.
Which of the following compounds is used to prepare orlon?
Which among the following polymers is obtained from CH2 = CH – CN by polymerisation?
Which of the following polymers is prepared by using phenol?
Which among the following polymers is an example of addition polymer?
Which of the following polymers is used as insulation for cables?
Match the polymers given in Column I with their repeating units given in Column II.
| Column I | Column II |
| (i) Acrilan |
(a) \[\begin{array}{cc} |
| (ii) Polystyrene | (b) \[\begin{array}{cc} \ce{Cl}\phantom{.......}\\ |\phantom{........}\\ \phantom{}\ce{-(CH2 - C = CH - CH2)\underset{n}{-}} \end{array}\] |
| (iii) Neoprene | (c) \[\begin{array}{cc} \phantom{................................}\ce{CN}\\ \phantom{..............................}|\\ \ce{-(CH2 - CH = CH - CH2 - CH2 - CH)\underset{n}{-}} \end{array}\] |
| (iv) Novolac | (d) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{.......}\ce{CN} \end{array}\] |
| (v) Buna—N | (e)  |
| (f) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{......}\ce{Cl} \end{array}\] |
Phenol and formaldehyde undergo condensation to give a polymar (A) which on heating with formaldehyde gives a thermosetting polymer (B). Name the polymers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers?
Which of the following is not a copolymer?
Which of the following products is formed when benzaldehyde is treated with CH3MgBr and the addition product so obtained is subjected to acid hydrolysis?
Which of the following is an example of polyester?
Which of the following polymer is used for manufacturing of buckets, dustbins, pipes, etc?
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
Name and draw the structure of the repeating unit in natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Match the following pairs:
| Polymer | Monomer | ||
| (i) | Teflon | (a) | CH2 = CH2 |
| (ii) | PVC | (b) | CF2 = CF2 |
| (iii) | Natural rubber | (c) | Isoprene |
| (iv) | Polythene | (d) | H2C=CHCl |
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Which from following statements is NOT true regarding neoprene?
