Advertisements
Advertisements
प्रश्न
Explain how and why will the rate of reaction for a given reaction be affected when the temperature at which the reaction was taking place is decreased.
Advertisements
उत्तर
The rate of reaction will decrease. At lower temperatures, the kinetic energy of molecules decreases thereby the collisions decrease resulting in a lowering of the rate of reaction.
APPEARS IN
संबंधित प्रश्न
Explain a graphical method to determine activation energy of a reaction.
What is the effect of adding a catalyst on Activation energy (Ea)
Predict the main product of the following reactions: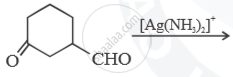
For an endothermic reaction energy of activation is Ea and enthalpy of reaction ΔH (both of there in KJ moI–1) minimum value of Ea will be ______.
The activation energy in a chemical reaction is defined as ______.
The slope of Arrhenius Plot `("In" "k" "v"//"s" 1/"T")` of first-order reaction is −5 × 103 K. The value of Ea of the reaction is. Choose the correct option for your answer. [Given R = 8.314 JK−1mol−1]
Arrhenius equation can be represented graphically as follows:

The (i) intercept and (ii) slope of the graph are:
The decomposition of N2O into N2 and O2 in the presence of gaseous argon follows second-order kinetics, with k = (5.0 × 1011 L mol−1 s−1) `"e"^(-(29000 "K")/"T")`. Arrhenius parameters are ______ kJ mol−1.
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
The rate of a reaction quadruples when temperature changes from 27°C to 57°C calculate the energy of activation.
(Given: R = 8. 314 J K−1 mol−1, log 4 = 0.6021)
