Advertisements
Advertisements
प्रश्न
Arrhenius equation can be represented graphically as follows:

The (i) intercept and (ii) slope of the graph are:
विकल्प
(i) ln A (ii) Ea/R
(i) A (ii) Ea
(i) ln A (ii) - Ea/R
(i) A (ii) - Ea
Advertisements
उत्तर
(i) ln A (ii) - Ea/R
Explanation:
According to Arrhenius equations,
`k = A exp (- (Ea)/(RT))`
Taking ln on both sides
`l n k = l n A - (Ea)/(RT)`
Or `l n K = (- (Ea)/R) 1/T + l n A`
Comparing the above equations with the equation of straight line y = mx + C, we got
m = `(-Ea)/R`
C = `l n A`
APPEARS IN
संबंधित प्रश्न
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581 K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
The rate constant of a first order reaction are 0.58 S-1 at 313 K and 0.045 S-1 at 293 K. What is the energy of activation for the reaction?
Predict the main product of the following reactions: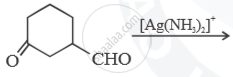
Which of the following statements are in accordance with the Arrhenius equation?
(i) Rate of a reaction increases with increase in temperature.
(ii) Rate of a reaction increases with decrease in activation energy.
(iii) Rate constant decreases exponentially with increase in temperature.
(iv) Rate of reaction decreases with decrease in activation energy.
Why does the rate of a reaction increase with rise in temperature?
What happens to most probable kinetic energy and the energy of activation with increase in temperature?
The slope of Arrhenius Plot `("In" "k" "v"//"s" 1/"T")` of first-order reaction is −5 × 103 K. The value of Ea of the reaction is. Choose the correct option for your answer. [Given R = 8.314 JK−1mol−1]
The equation k = `(6.5 xx 10^12 "s"^(-1))"e"^(- 26000 " K"//"T")` is followed for the decomposition of compound A. The activation energy for the reaction is ______ kJ mol-1. (Nearest integer) (Given: R = 8.314 JK-1 mol-1)
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below

The reaction must be:
Activation energy of any chemical reactions can be calculated if one knows the value of:
