Advertisements
Advertisements
प्रश्न
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
Advertisements
उत्तर
As the temperature of a chemical reaction rises, the rate constant k rises and the activation energy Ea falls.
According to Arrhenius equation
k = `"Ae"^(-"E"_"a"//"RT")`
As a result, the rate constant k rises exponentially as the temperature rises.
APPEARS IN
संबंधित प्रश्न
The rate constant of a first order reaction increases from 4 × 10−2 to 8 × 10−2 when the temperature changes from 27°C to 37°C. Calculate the energy of activation (Ea). (log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
What will be the effect of temperature on rate constant?
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581 K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
Consider a certain reaction \[\ce{A -> Products}\] with k = 2.0 × 10−2 s−1. Calculate the concentration of A remaining after 100 s if the initial concentration of A is 1.0 mol L−1.
Define activation energy.
Predict the main product of the following reactions: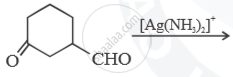
Which of the following graphs represents exothermic reaction?
(a)

(b)
(c)
The reaction between \[\ce{H2(g)}\] and \[\ce{O2(g)}\] is highly feasible yet allowing the gases to stand at room temperature in the same vessel does not lead to the formation of water. Explain.
Explain how and why will the rate of reaction for a given reaction be affected when the temperature at which the reaction was taking place is decreased.
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below

The reaction must be:
