Advertisements
Advertisements
प्रश्न
Draw neat labelled diagrams for two different experiments to prove that – hydrogen is lighter than air.
Advertisements
उत्तर
Experiment 1
Procedure: Bubble hydrogen gas by means of a small pipe into a dish containing soap solution.
Observation: Soap bubbles filled with H2 rise upwards.
Conclusion: Hydrogen is lighter than air hence soap bubbles filled with hydrogen rise up.

Experiment 2
Procedure: Take two dry test tubes.
Test tube 'A' - Filled with hydrogen.
Test tube 'B' - Filled with air.
Place test tube 'B' over test tube 'A' and test the gas in the upper test tube 'B' with a glowing splinter.
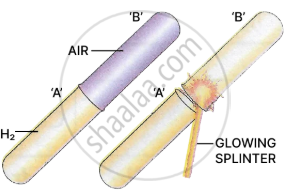
Observation: A 'pop' sound is heard in test tube 'B'. [hydrogen burns with a 'pop' sound].
Conclusion: Hydrogen is lighter than air and has moved upwards displacing the air.
APPEARS IN
संबंधित प्रश्न
Indicate which of the following statement is true and which is false:
Zinc can liberate hydrogen from water, acid and alkali solution.
Name the chemicals required to prepare hydrogen gas in the laboratory.
Draw a neat and well-labelled diagram for the laboratory preparation of hydrogen.
How is hydrogen gas collected? Why?
Multiple Choice Question
In metal activity series the more reactive metals are at
Give a test to identify hydrogen ?
Name the impurities present in hydrogen prepared in the laboratory.
How can these impurities be removed?
How would you show that hydrogen is lighter than air?
Starting from zinc how would you obtain hydrogen using a dilute acid.
[Give balanced equation & name the product formed in the case other than hydrogen].
Name a metal which will not react with the reactants above to give hydrogen.
Give reason for the following:
In the laboratory preparation of hydrogen from zinc and dilute hydrochloric acid – the zinc used granulated zinc.
Give a balanced equation for the following conversions sodium plumbite from lead.
In the laboratory preparation of hydrogen from zinc and dil. acid. Give a reason for the following:
The complete apparatus is air-tight.
In the laboratory preparation of hydrogen from zinc and dil. acid. Give a reason for the following:
Dilute nitric acid is not preferred as the reactant acid.
Give a reason for the following.
Nitric acid in the dilute form is not used in the laboratory preparation of hydrogen from metals.
Name the following.
A gaseous reducing agent which is basic in nature.
Select the correct answer from the symbol in the bracket.
The element which forms a diatomic molecule.
The diagram represent the preparation and collection of hydrogen by a standard
laboratory method.

State what is added through the thistle funnel ‘Y’
The diagram represent the preparation and collection of hydrogen by a standard laboratory method.

Name a gas other than hydrogen collected by the same method.
