Advertisements
Advertisements
प्रश्न
Draw neat labelled diagrams for two different experiments to prove that – hydrogen is lighter than air.
Advertisements
उत्तर
Experiment 1
Procedure: Bubble hydrogen gas by means of a small pipe into a dish containing soap solution.
Observation: Soap bubbles filled with H2 rise upwards.
Conclusion: Hydrogen is lighter than air hence soap bubbles filled with hydrogen rise up.

Experiment 2
Procedure: Take two dry test tubes.
Test tube 'A' - Filled with hydrogen.
Test tube 'B' - Filled with air.
Place test tube 'B' over test tube 'A' and test the gas in the upper test tube 'B' with a glowing splinter.
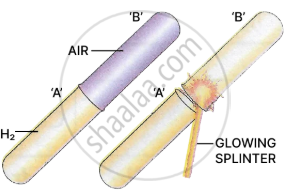
Observation: A 'pop' sound is heard in test tube 'B'. [hydrogen burns with a 'pop' sound].
Conclusion: Hydrogen is lighter than air and has moved upwards displacing the air.
APPEARS IN
संबंधित प्रश्न
Indicate which of the following statement is true and which is false:
Nitric acid cannot be used to prepare hydrogen by its action on active metals.
Draw a neat and well-labelled diagram for the laboratory preparation of hydrogen.
How would you show that hydrogen is a non-supporter of combustion?
FILL IN THE BLANK
........................ zinc is preferred over pure zinc in the laboratory preparation of hydrogen.
Give a test to identify hydrogen ?
How can these impurities be removed?
Give reason:
Apparatus for laboratory preparation of hydrogen should be airtight and away from a naked flame.
Starting from zinc how would you obtain hydrogen using a dilute acid.
[Give balanced equation & name the product formed in the case other than hydrogen].
Name a metal which will not react with the reactants above to give hydrogen.
Give balanced equation for the following conversion:
Zinc to sodium zincate – using an alkali.
Give balanced equation for the following conversion:
Acidified water to hydrogen – by electrolysis.
Give reason for the following:
In the laboratory preparation of hydrogen from zinc and dilute hydrochloric acid – the zinc used granulated zinc.
Complete and balance the equation:
[Laboratory method]
By action of dilute acid on zinc
Zinc - Zn + 2HCl → _____ + _____ [g]
Give a balanced equation for the following conversions sodium plumbite from lead.
Give a balanced equation for the following conversions sodium aluminate from aluminium.
In the laboratory preparation of hydrogen from zinc and dil. acid. Give a reason for the following:
The complete apparatus is air-tight.
In the laboratory preparation of hydrogen from zinc and dil. acid. Give a reason for the following:
Hydrogen is not collected over air.
Name the following.
A gaseous reducing agent which is basic in nature.
Select the correct answer from the symbol in the bracket.
The element which forms a diatomic molecule.
The diagram represent the preparation and collection of hydrogen by a standard laboratory method.

State, why hydrogen is collected after all the air in the apparatus, is allowed to escape.
