Advertisements
Advertisements
प्रश्न
Balance the following reaction by oxidation number method.
\[\ce{H2SO4_{(aq)} + C_{(s)} -> CO2_{(g)} + SO2_{(g)} + H2O_{(l)}(acidic)}\]
Advertisements
उत्तर
\[\ce{H2SO4_{(aq)} + C_{(s)} -> CO2_{(g)} + SO2_{(g)} + H2O_{(l)}(acidic)}\]
Step 1: Write the skeletal equation and balance the elements other than O and H.
\[\ce{H2SO4_{(aq)} + C_{(s)} -> CO2_{(g)} + SO2_{(g)} + H2O_{(l)}}\]
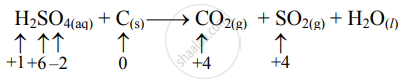
Step 2: Assign oxidation numbers to S and C. Calculate the increase and decrease in the oxidation number and make them equal.
Increase in oxidation number:

(Increase per atom = 4)
Decrease in oxidation number:
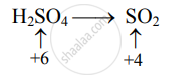
(Decrease per atom = 2)
To make the net increase and decrease equal, we must take 2 atoms of S.
\[\ce{2H2SO4_{(aq)} + C_{(s)} -> CO2_{(g)} + 2SO2_{(g)} + H2O_{(l)}}\]
Step 3: Balance ‘O’ atoms by adding H2O to the right-hand side.
\[\ce{2H2SO4_{(aq)} + C_{(s)} -> CO2_{(g)} + 2SO2_{(g)} + H2O_{(l)} + H2O_{(l)}}\]
Step 4: The medium is acidic. There is no charge on either side. Hydrogen atoms are equal on both sides.
\[\ce{2H2SO4_{(aq)} + C_{(s)} -> CO2_{(g)} + 2SO2_{(g)} + 2H2O_{(l)}}\]
Step 5: Check two sides for the balance of atoms and charges.
Hence, balanced equation is \[\ce{2H2SO4_{(aq)} + C_{(s)} -> CO2_{(g)} + 2SO2_{(g)} + 2H2O_{(l)}}\]
APPEARS IN
संबंधित प्रश्न
The compound AgF2 is an unstable compound. However, if formed, the compound acts as a very strong oxidizing agent. Why?
How do you count for the following observations?
Though alkaline potassium permanganate and acidic potassium permanganate both are used as oxidants, yet in the manufacture of benzoic acid from toluene we use alcoholic potassium permanganate as an oxidant. Why? Write a balanced redox equation for the reaction.
Balance the following equation in the basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{N2H4(l) + ClO^-_3 (aq) → NO(g) + Cl–(g)}\]
Balance the following equation in basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{Cl_2O_{7(g)} + H_2O_{2(aq)} -> ClO-_{2(aq)} + O_{2(g)} + H+_{(aq)}}\]
Chlorine is used to purify drinking water. Excess of chlorine is harmful. The excess of chlorine is removed by treating with sulphur dioxide. Present a balanced equation for this redox change taking place in water.
In Ostwald’s process for the manufacture of nitric acid, the first step involves the oxidation of ammonia gas by oxygen gas to give nitric oxide gas and steam. What is the maximum weight of nitric oxide that can be obtained starting only with 10.00 g. of ammonia and 20.00 g of oxygen?
Choose the correct option.
For the following redox reactions, find the correct statement.
\[\ce{Sn^{2⊕} + 2Fe^{3⊕}->Sn^{4⊕} + 2Fe^{2⊕}}\]
Justify that the following reaction is redox reaction; identify the species oxidized/reduced, which acts as an oxidant and which acts as a reductant.
\[\ce{2Cu2O_{(S)} + Cu2S_{(S)}->6Cu_{(S)} + SO2_{(g)}}\]
Balance the following reaction by oxidation number method.
\[\ce{Cr2O^2-_{7(aq)} + SO^2-_{3(aq)}->Cr^3+_{ (aq)} + SO^2-_{4(aq)}(acidic)}\]
Balance the following reaction by oxidation number method.
\[\ce{MnO^-_{4(aq)} + Br^-_{ (aq)}->MnO2_{ (s)} + BrO^-_{3(aq)}(basic)}\]
Balance the following redox equation by half-reaction method.
\[\ce{H2C2O_{4(aq)} + MnO^-_{4(aq)}->CO2_{(g)} + Mn^2+_{( aq)}(acidic)}\]
What is the change in oxidation number of Sulphur in following reaction?
\[\ce{MnO^-_{4(aq)} + SO^{2-}_{3(aq)} -> MnO^{2-}_{4(aq)} + SO^{2-}_{4(aq)}}\]
Write balanced chemical equation for the following reactions:
Permanganate ion \[\ce{(MnO^{-}4)}\] reacts with sulphur dioxide gas in acidic medium to produce \[\ce{Mn^{2+}}\] and hydrogen sulphate ion.
Write balanced chemical equation for the following reactions:
Dichlorine heptaoxide \[\ce{(Cl2O7)}\] in gaseous state combines with an aqueous solution of hydrogen peroxide in acidic medium to give chlorite ion \[\ce{(ClO^{-}2)}\] and oxygen gas. (Balance by ion-electron method)
Balance the following equations by the oxidation number method.
\[\ce{Fe^{2+} + H^{+} + Cr2O^{2-}7 -> Cr^{3+} + Fe^{3+} + H2O}\]
Balance the following equations by the oxidation number method.
\[\ce{I2 + S2O^{2-}3 -> I- + S4O^{2-}6}\]
Balance the following equations by the oxidation number method.
\[\ce{MnO2 + C2O^{2-}4 -> Mn^{2+} + CO2}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{3HCl (aq) + HNO3 (aq) -> Cl2 (g) + NOCl (g) + 2H2O (l)}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{HgCl2 (aq) + 2KI (aq) -> HgI2 (s) + 2KCl (aq)}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{4NH3 (g) + 3O2 (g) -> 2N2 (g) + 6H2O (g)}\]
Balance the following ionic equations.
\[\ce{Cr2O^{2-}7 + Fe^{2+} + H+ -> Cr^{3+} + Fe^{3+} + H2O}\]
Balance the following ionic equations.
\[\ce{MnO^{-}4 + SO^{2-}3 + H^{+} -> Mn^{2+} + SO^{2-}4 + H2O}\]
Balance the following ionic equations.
\[\ce{MnO^{-}4 + H^{+} + Br^{-} -> Mn^{2+} + Br2 + H2O}\]
In \[\ce{Cu^{2+} + Ag -> Cu + Ag^+}\], oxidation half-reaction is:
The weight of CO is required to form Re2(CO)10 will be ______ g, from 2.50 g of Re2O7 according to given reaction
\[\ce{Re2O7 + CO -> Re2(CO)10 + CO2}\]
Atomic weight of Re = 186.2; C = 12 and O = 16.
\[\ce{H2O2 -> 2H^+ + O2 + 2e^-}\]; E0 = −0.68 V.
This equation represents which of the following behaviour of H2O2?
