Topics
Circular Motion
- Angular Displacement
- Angular Velocity
- Angular Acceleration
- Angular Velocity and Its Relation with Linear Velocity
- Uniform Circular Motion (UCM)
- Radial Acceleration
- Dynamics of Uniform Circular Motion - Centripetal Force
- Centrifugal Forces
- Banking of Roads
- Vertical Circular Motion Due to Earth’s Gravitation
- Equation for Velocity and Energy at Different Positions of Vertical Circular Motion
- Kinematical Equations for Circular Motion in Analogy with Linear Motion.
Rotational Dynamics
- Rotational Dynamics
- Circular Motion and Its Characteristics
- Applications of Uniform Circular Motion
- Vertical Circular Motion
- Moment of Inertia as an Analogous Quantity for Mass
- Radius of Gyration
- Theorems of Perpendicular and Parallel Axes
- Angular Momentum or Moment of Linear Momentum
- Expression for Torque in Terms of Moment of Inertia
- Conservation of Angular Momentum
- Rolling Motion
- Overview: Rotational Dynamics
Gravitation
- Newton’s Law of Gravitation
- Periodic Time
- Kepler’s Laws
- Binding Energy and Escape Velocity of a Satellite
- Weightlessness
- Variation of ‘G’ Due to Lattitude and Motion
- Variation in the Acceleration>Variation in Gravity with Altitude
- Communication satellite and its uses
- Composition of Two S.H.M.’S Having Same Period and Along Same Line
Mechanical Properties of Fluids
- Fluid and Its Properties
- Thrust and Pressure
- Pressure of liquid
- Pressure Exerted by a Liquid Column
- Atmospheric Pressure
- Gauge Pressure and Absolute Pressure
- Hydrostatic Paradox
- Pascal’s Law
- Application of Pascal’s Law
- Measurement of Atmospheric Pressure
- Mercury Barometer (Simple Barometer)
- Open Tube Manometer
- Surface Tension
- Molecular Theory of Surface Tension
- Surface Tension and Surface Energy
- Angle of Contact
- Effect of Impurity and Temperature on Surface Tension
- Excess Pressure Across the Free Surface of a Liquid
- Explanation of Formation of Drops and Bubbles
- Capillarity and Capillary Action
- Fluids in Motion
- Critical Velocity and Reynolds Number
- Viscous Force or Viscosity
- Stokes’ Law
- Terminal Velocity
- Continuous and Discontinuous Functions
- Bernoulli's Equation
- Applications of Bernoulli’s Equation
- Overview: Mechanical Properties of Fluids
Kinetic Theory of Gases and Radiation
- Gases and Its Characteristics
- Classification of Gases: Real Gases and Ideal Gases
- Mean Free Path
- Expression for Pressure Exerted by a Gas
- Root Mean Square (RMS) Speed
- Interpretation of Temperature in Kinetic Theory
- Law of Equipartition of Energy
- Specific Heat Capacity
- Absorption, Reflection, and Transmission of Heat Radiation
- Perfect Blackbody
- Emission of Heat Radiation
- Kirchhoff’s Law of Heat Radiation and Its Theoretical Proof
- Spectral Distribution of Blackbody Radiation
- Wien's Displacement Law
- Stefan-boltzmann Law of Radiation
- Overview: Kinetic Theory of Gases and Radiation
Angular Momentum
- Definition of M.I., K.E. of Rotating Body
- Rolling Motion
- Physical Significance of M.I (Moment of Inertia)
- Torque and Angular Momentum
- Theorems of Perpendicular and Parallel Axes
- M.I. of Some Regular Shaped Bodies About Specific Axes
Thermodynamics
- Thermodynamics
- Thermal Equilibrium
- Measurement of Temperature
- Heat, Internal Energy and Work
- First Law of Thermodynamics
- Thermodynamic State Variables and Equation of State
- Thermodynamic Process
- Heat Engine
- Refrigerators and Heat Pumps
- Entropy and Second Law of Thermodynamics
- Carnot Cycle and Carnot Engine
- Overview: Thermodynamics
Oscillations
- Periodic and Oscillatory Motion
- Simple Harmonic Motion (S.H.M.)
- Differential Equation of Linear S.H.M.
- Projection of U.C.M.(Uniform Circular Motion) on Any Diameter
- Phase of K.E (Kinetic Energy)
- K.E.(Kinetic Energy) and P.E.(Potential Energy) in S.H.M.
- Composition of Two S.H.M.’S Having Same Period and Along Same Line
- Some Systems Executing Simple Harmonic Motion
Oscillations
- Oscillations
- Explanation of Periodic Motion
- Linear Simple Harmonic Motion (S.H.M.)
- Differential Equation of Linear S.H.M.
- Acceleration (a), Velocity (v) and Displacement (x) of S.H.M.
- Amplitude (A), Period (T) and Frequency (N) of S.H.M.
- Reference Circle Method
- Phase in S.H.M.
- Graphical Representation of S.H.M.
- Composition of Two S.H.M.’S Having Same Period and Along Same Line
- The Energy of a Particle Performing S.H.M.
- Simple Pendulum
- Angular S.H.M. and It's Differential Equation
- Damped Oscillations
- Free Oscillations, Forced Oscillations and Resonance Oscillations
- Periodic and Oscillatory Motion
- Overview: Oscillations
Elasticity
- Eneral Explanation of Elastic Property
- Stress and Strain
- Hooke’s Law
- Elastic Energy
- Elastic Constants and Their Relation
- Determination of ‘Y’
- Behaviour of Metal Wire Under Increasing Load
- Application of Elastic Behaviour of Materials
Surface Tension
- Molecular Theory of Surface Tension
- Surface Tension
- Capillarity and Capillary Action
- Effect of Impurity and Temperature on Surface Tension
Superposition of Waves
Wave Motion
- Wave Motion Introduction
- Simple Harmonic Progressive Waves,
- Reflection of Transverse and Longitudinal Waves
- Change of Phase
- Principle of Superposition of Waves
- Formation of Beats
- Beats
Wave Optics
- Concept of Wave Optics
- Nature of Light
- Light as a Wave
- Huygens’ Theory
- Reflection of Light at a Plane Surface
- Refraction of Light at a Plane Boundary Between Two Media
- Polarisation of Light
- Interference
- Diffraction of Light
- Resolving Power
- Overview: Wave Optics
Stationary Waves
- Study of Vibrations in a Finite Medium
- Formation of Stationary Waves on String
- Study of Vibrations of Air Columns
- Free and Forced Vibrations
- Forced Oscillations and Resonance
Electrostatics
- Concept of Electrostatics
- Application of Gauss' Law
- Electric Potential and Potential Difference
- Electric Potential Due to a Point Charge
- Equipotential Surfaces
- Electrical Energy of Two Point Charges and of a Dipole in an Electrostatic Field
- Free Charges and Bound Charges Inside a Conductor
- Combination of Capacitors
- Displacement Current
- Energy Stored in a Charged Capacitor
- Van De Graaff Generator
- Uniformly Charged Infinite Plane Sheet and Uniformly Charged Thin Spherical Shell (Field Inside and Outside)
- Overview: Electrostatics
Kinetic Theory of Gases and Radiation
- Concept of an Ideal Gas
- Assumptions of Kinetic Theory of Gases
- Derivation for Pressure of a Gas
- Degrees of Freedom
- Derivation of Boyle’s Law
- Thermal Equilibrium
- First Law of Thermodynamics
- Heat Engine
- Temperature and Heat
- Qualitative Ideas of Black Body Radiation
- Wien's Displacement Law
- Green House Effect
- Stefan's Law
- Maxwell Distribution
- Specific Heat Capacities - Gases
- Law of Equipartition of Energy
Current Electricity
Wave Theory of Light
Magnetic Fields Due to Electric Current
- Magnetic Fields Due to Electric Current
- Magnetic force
- Cyclotron
- Helical Motion
- Magnetic Force on a Wire Carrying a Current
- Force on a Closed Circuit in a Magnetic Field
- Torque on a Current-Loop in a Uniform Magnetic Field
- Magnetic Dipole Moment
- Magnetic Potential Energy of a Dipole
- Biot-Savart Law
- Force of Attraction Between Two Long Parallel Wires
- Magnetic Field Produced by a Current in a Circular Arc of a Wire
- Magnetic Field on the Axis of a Circular Current-Carrying Loop
- Magnetic Lines for a Current Loop
- Ampere’s Circuital Law
- Applications of Ampere’s Circuital Law > Magnetic Field of a Toroidal Solenoid
- Overview: Magnetic Fields Due to Electric Current
Magnetic Materials
- Magnetic Materials
- Torque Acting on a Magnetic Dipole in a Uniform Magnetic Field
- Origin of Magnetism in Materials
- Magnetisation and Magnetic Intensity
- Magnetic Properties of Materials
- Classification of Magnetic Materials
- Hysteresis: Retentivity and Coercivity
- Permanent Magnet
- Magnetic Shielding
- Overview: Magnetic Materials
Interference and Diffraction
- Interference of Light
- Conditions for Producing Steady Interference Pattern
- Interference of Light Waves and Young’s Experiment
- Analytical Treatment of Interference Bands
- Measurement of Wavelength by Biprism Experiment
- Fraunhofer Diffraction Due to a Single Slit
- Rayleigh’s Criterion
- Resolving Power of a Microscope and Telescope
- Difference Between Interference and Diffraction
Electrostatics
- Mechanical Force on Unit Area of a Charged Conductor
- Energy Density of a Medium
- Concept of Condenser
- The Parallel Plate Capacitor
- Capacity of Parallel Plate Condenser
- Effect of Dielectric on Capacitance
- Energy of Charged Condenser
- Condensers in Series and Parallel,
- Van-deGraaff Generator
Electromagnetic Induction
- Electromagnetic Induction
- Faraday's Laws of Electromagnetic Induction
- Lenz's Law
- Flux of the Field
- Motional Electromotive Force (e.m.f.)
- Induced Emf in a Stationary Coil in a Changing Magnetic Field
- Generators
- Back Emf and Back Torque
- Induction and Energy Transfer
- Eddy Currents or Foucault Currents
- Self Inductance
- Energy Stored in a Magnetic Field
- Energy Density of a Magnetic Field
- Mutual Inductance
- Transformers
- Overview of Electromagnetic Induction
Current Electricity
- Meter Bridge
AC Circuits
- AC Circuits
- Values of Alternating Current
- Phasors
- AC Voltage Applied to a Resistor
- AC Voltage Applied to an Inductor
- AC Voltage Applied to a Capacitor
- AC Voltage Applied to a Series LCR Circuit
- Power in AC Circuit
- LC Oscillations
- Electric Resonance
- Sharpness of Resonance: Q Factor
- Choke Coil
- Overview: AC Circuits
Dual Nature of Radiation and Matter
- Dual Nature of Radiation and Matter
- The Photoelectric Effect
- Wave-particle Duality of Electromagnetic Radiation
- Photo Cell
- De Broglie Hypothesis
- Davisson and Germer Experiment
- Wave-particle Duality of Matter
- Overview: Dual Nature of Radiation and Matter
Magnetic Effects of Electric Current
Structure of Atoms and Nuclei
- Structure of the Atom and Nucleus
- Thomson’s Atomic Model
- Geiger-marsden Experiment
- Lord Rutherford’s Atomic model
- Atomic Spectra
- Neils Bohr’s Model of an Atom
- Atomic Nucleus
- Constituents of a Nucleus
- Isotopes
- Atomic and Nuclear Masses
- Size and Density of the Nucleus
- Mass Defect and Binding Energy
- Binding Energy Curve
- Forms of Energy > Nuclear Energy
- Nuclear Binding Energy
- Radioactive Decays
- Law of Radioactive Decay
- Overview: Structure of Atoms and Nuclei
Magnetism
Electromagnetic Inductions
- Electromagnetic Induction
- Self Inductance
- Mutual Inductance
- Transformers
- Need for Displacement Current
- Coil Rotating in Uniform Magnetic Induction
- A.C. Generator
- Reactance and Impedance
- LC Oscillations
- Inductance and Capacitance
- Resonant Circuits
- Power in AC Circuit
- Lenz’s Law and Conservation of Energy
Semiconductor Devices
Electrons and Photons
Atoms, Molecules and Nuclei
- Alpha-particle Scattering and Rutherford’s Nuclear Model of Atom
- Bohr’s Model for Hydrogen Atom
- Hydrogen Spectrum
- Atomic Masses and Composition of Nucleus
- Radioactivity
- Law of Radioactive Decay
- Atomic Mass, Mass - Energy Relation and Mass Defect
- Nuclear Binding Energy
- Nuclear Fusion
- de-Broglie Relation
- Wave Nature of Matter
- Wavelength of an Electron
- Davisson and Germer Experiment
- Continuous and Characteristics X-rays
- Mass Defect and Binding Energy
Semiconductors
- Energy Bands in Solids
- Extrinsic Semiconductor
- Applications of n-type and p-type Semiconductors
- Special Purpose P-n Junction Diodes
- Semiconductor Diode
- Voltage Regulator
- I-V Characteristics of Led
- Transistor and Characteristics of a Transistor
- Transistor as an Amplifier (Ce-configuration)
- Transistor as a Switch
- Oscillators
- Digital Electronics and Logic Gates
Communication Systems
Definition: Binding Energy
The energy equivalent to that of mass defect, i.e., the energy required for holding the nucleons together in a nucleus, is called the Binding Energy of the nucleus.
Definition: Binding Energy Per Nucleon
The ratio of the binding energy \[E_n\] of a nucleus to the number of nucleons A in that nucleus is called Binding Energy Per Nucleon.
Definition: Energy Levels
The definite amount of energies associated with the electrons in different orbits of an atom are called the Energy Levels (of that atom).
Definition: Excitation Energy
The energy required to take an electron from the ground state to an excited state is called the Excitation Energy of the electron in that state.
Definition: Binding Energy of Electron
The minimum energy required to make an electron free from the nucleus is called the Binding Energy of an electron.
Definition: Ionization Energy
The minimum amount of energy required to be given to an electron in the ground state of an atom to set the electron free is called the Ionization Energy of that atom.
Formula: Binding Energy
Eb = ΔM ⋅ c2
Eb = [(Zmp + (A − Z)mn) − M] × c2
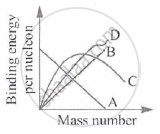
Definition: Binding Energy Curve
In a graph plotting binding energy per nucleon (Bₙ) against mass number (A) for all known nuclei, the resulting curve is called binding energy curve.
Maharashtra State Board: Class 11
Formula: Binding Energy per Nucleon
BE per nucleon = \[\frac {E.E.}{A}\]
Formula: Binding Energy from Mass Defect
Binding Energy = \[(\Delta m)\cdot c^2=(\text{Mass defect})\cdot c^2\]
Formula: Binding Energy per Nucleon (from Nucleon Number)
\[\text{Binding Energy per Nucleon}=\frac{\text{Binding Energy}}{\text{Nucleon Number}}\]
Key Points: Binding Energy per Nucleon
- The greater the binding energy per nucleon, the more stable the nucleus.
- Iron-56 (Fe⁵⁶) and Nickel-62 are among the most stable nuclei, lying at the peak of the binding energy curve.
- Light nuclei (A < 20): Binding energy per nucleon increases rapidly with mass number.
- Intermediate nuclei (A ≈ 20–60): Highest binding energy per nucleon — most stable region.
- Heavy nuclei (A > 60): Binding energy per nucleon gradually decreases — less tightly bound.
- Very heavy nuclei can become unstable and may undergo fission, splitting into smaller, more stable nuclei, releasing energy.
- If nucleons are separated, the energy required to separate them gets converted into mass.