Advertisements
Advertisements
What is a cell constant? What are its units? How is it determined experimentally?
Concept: undefined >> undefined
Answer the following in one or two sentences.
Write the relationship between conductivity and molar conductivity and hence unit of molar conductivity.
Concept: undefined >> undefined
Advertisements
Answer the following in brief.
Explain the effect of dilution of the solution on conductivity.
Concept: undefined >> undefined
Answer the following:
What is Kohlrausch law of independent migration of ions? How is it useful in obtaining molar conductivity at zero concentration of a weak electrolyte? Explain with an example.
Concept: undefined >> undefined
Answer the following.
The first ionization enthalpies of S, Cl, and Ar are 1000, 1256 and 1520 kJ/mol-1, respectively. Explain the observed trend.
Concept: undefined >> undefined
Explain the trend in following atomic properties of group 16 elements:
Atomic radii
Concept: undefined >> undefined
Answer the following.
Explain the trend in following atomic properties of group 16 elements:
Ionisation enthalpy
Concept: undefined >> undefined
Explain the trend in following atomic properties of group 16 elements:
Electronegativity
Concept: undefined >> undefined
Choose the most correct option.
Electronic configuration of Cu and Cu1+ are _______.
Concept: undefined >> undefined
Choose the most correct option.
Which of the following have d0s0 configuration?
Concept: undefined >> undefined
Choose the most correct option.
In which of the following series all the elements are radioactive in nature?
Concept: undefined >> undefined
Answer the following
Write the probable electronic configuration of chromium and copper.
Concept: undefined >> undefined
Choose the most correct option.
The oxidation state of cobalt ion in the complex [Co(NH3)5 Br]SO4 is __________.
Concept: undefined >> undefined
Answer the following in one or two sentences.
What is the shape of a complex in which the coordination number of the central metal ion is 4?
Concept: undefined >> undefined
Write the structure and IUPAC name of the major product in the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH2Cl + NaI ->[Acetone]}\\
|\phantom{..................}\\
\ce{CH3\phantom{...............}}
\end{array}\]
Concept: undefined >> undefined
Write the structure and IUPAC name of the major product in the following reaction.
\[\ce{CH3 - CH2Br + SbF3 ->}\]
Concept: undefined >> undefined
Write the structure and IUPAC name of the major product in the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH = CH2 + HBr ->[peroxide]}\\
|\phantom{........................}\\
\ce{CH3\phantom{.....................}}
\end{array}\]
Concept: undefined >> undefined
Write the structure and IUPAC name of the major product in the following reaction.

Concept: undefined >> undefined
Write the structure and IUPAC name of the major product in the following reaction.
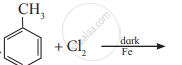
Concept: undefined >> undefined
Give reason:
Alkyl halides are generally not prepared by free radical halogenation of alkanes.
Concept: undefined >> undefined
