Please select a subject first
Advertisements
Advertisements
Write true or false. Correct the false statement.
Free radicals are negatively charged species.
Concept: undefined >> undefined
Write true or false. Correct the false statement.
Aniline is a heterocyclic compound.
Concept: undefined >> undefined
Advertisements
Draw all the no-bond resonance structures of isopropyl carbocation.
Concept: undefined >> undefined
Answer the following.
What is the hybridization of Al in AlCl3?
Concept: undefined >> undefined
Answer the following.
Name a molecule having a banana bond.
Concept: undefined >> undefined
Draw the structure of the following.
Orthophosphoric acid
Concept: undefined >> undefined
Draw the structure of the following.
Resonance structure of nitric acid
Concept: undefined >> undefined
Choose the correct option.
Which of the following statements are true with respect to electronic displacement in a covalent bond?
a. Inductive effect operates through π bond
b. Resonance effect operates through σ bond
c. Inductive effect operates through σ bond
d. Resonance effect operates through π bond
Concept: undefined >> undefined
Choose the correct option.
Hyperconjugation involves overlap of ______ orbitals.
Concept: undefined >> undefined
The correct IUPAC name of the compound 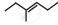 is ______.
is ______.
Concept: undefined >> undefined
Choose the correct option.
The geometry of a carbocation is ______.
Concept: undefined >> undefined
Choose the correct option.
The homologous series of alcohols has general molecular formula ______.
Concept: undefined >> undefined
Choose the correct option.
The delocalization of electrons due to overlap between p orbital and sigma bond is called _______.
Concept: undefined >> undefined
Explain the structure and bonding of diborane.
Concept: undefined >> undefined
Match the pairs from columns A and B.
| column A | column B |
|
i) BCl3 |
a. Angular molecule |
| ii) SiO2 | b. linear covalent molecule |
| iii. CO2 | c. Tetrahedral molecule |
| d. Planar trigonal molecule |
Concept: undefined >> undefined
State true or false. Correct the false statement.
The tendency for catenation is much higher for C than for Si.
Concept: undefined >> undefined
Shravani was performing inorganic qualitative analysis of a salt. To an aqueous solution of that salt, she added silver nitrate. When a white precipitate was formed. On adding ammonium hydroxide to this, she obtained a clear solution. Comment on her observations and write the chemical reactions involved.
Concept: undefined >> undefined
Write Postulates of the kinetic theory of gases.
Concept: undefined >> undefined
A compound is prepared from the mineral colemanite by boiling it with a solution of sodium carbonate. It is white crystalline solid and used for inorganic qualitative analysis.
Write the reaction that explains its formation.
Concept: undefined >> undefined
Shravani was performing inorganic qualitative analysis of a salt. To an aqueous solution of that salt, she added silver nitrate. When a white precipitate was formed. On adding ammonium hydroxide to this, she obtained a clear solution. Comment on her observations and write the chemical reactions involved.
Concept: undefined >> undefined
