Advertisements
Advertisements
If NaCl is doped with 10−3 mol % of SrCl2, what is the concentration of cation vacancies?
Concept: undefined >> undefined
Calculate the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 in 450 mL of water at 37°C.
Concept: undefined >> undefined
Advertisements
At 300 K, 36 g of glucose present in a litre of its solution has an osmotic pressure of 4.98 bar. If the osmotic pressure of the solution is 1.52 bars at the same temperature, what would be its concentration?
Concept: undefined >> undefined
Determine the amount of CaCl2 (i = 2.47) dissolved in 2.5 litre of water such that its osmotic pressure is 0.75 atm at 27°C.
Concept: undefined >> undefined
Determine the osmotic pressure of a solution prepared by dissolving 25 mg of K2SO4 in 2 liter of water at 25°C, assuming that it is completely dissociated.
Concept: undefined >> undefined
What is the difference between multimolecular and macromolecular colloids? Give one example of each. How are associated colloids different from these two types of colloids?
Concept: undefined >> undefined
What are micelles? Give an example of a micellers system.
Concept: undefined >> undefined
Calculate the overall complex dissociation equilibrium constant for the Cu(NH3)42+ ion, given that β4 for this complex is 2.1 × 1013.
Concept: undefined >> undefined
What is meant by stability of a coordination compound in solution?
Concept: undefined >> undefined
Where does the water present in the egg go after boiling the egg?
Concept: undefined >> undefined
What is the effect of denaturation on the structure of proteins?
Concept: undefined >> undefined
Write a short note on diazotisation.
Concept: undefined >> undefined
Give the IUPAC name of the following compound:
PhCH2CH2COOH
Concept: undefined >> undefined
Give the IUPAC name of the following compound:
(CH3)2C=CHCOOH
Concept: undefined >> undefined
Give the IUPAC name of the following compound:

Concept: undefined >> undefined
Give the IUPAC name of the following compound:
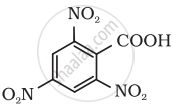
Concept: undefined >> undefined
Why carboxylic acid does not give reactions of carbonyl group?
Concept: undefined >> undefined
Illustrate the following reaction giving suitable example in each case: Diazotisation
Concept: undefined >> undefined
Following reactions occur at cathode during the electrolysis of aqueous silver chloride solution :
`Ag^+(aq)+e^(-)rarrAg(s)" "" "" "E^@=+0.80V`
`H^+(aq)+e^(-)rarr1/2H_2(g)" "" "" " E^@=0.00V`
On the basis of their standard reduction electrode potential (E°) values, which reaction is feasible at the cathode and why ?
Concept: undefined >> undefined
Define the following term :
Peptization
Concept: undefined >> undefined
