Please select a subject first
Advertisements
Advertisements
Write an equation for the reaction of hydrochloric acid on:
Manganese (IV) oxide
Concept: undefined >> undefined
Convert Hydrochloric acid to nascent chlorine.
Concept: undefined >> undefined
Advertisements
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NA4OH + HCl -> }\]
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.

Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilutes or conc. acid is used.
\[\ce{NH4OH + HCl -> }\]
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
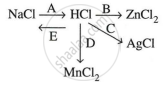
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl ->}\]
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH +HCI ->}\]
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
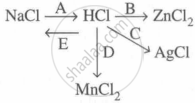
Concept: undefined >> undefined
Convert Hydrochloric acid to nascent chlorine.
Concept: undefined >> undefined
Identify the alloy in the given composition.
iron, nickel, chromium, carbon
Concept: undefined >> undefined
Write one use of the following alloy:
Bronze
Concept: undefined >> undefined
Write one use of the following alloy:
Fuse metal
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
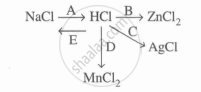
Concept: undefined >> undefined
Complete and balance the following reaction; state whether it is dilute or cone. acid is used.
\[\ce{NH_4OH + HCl ->}\]
Concept: undefined >> undefined
Convert hydrochloric acid to nascent chlorine.
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or conc. acid is used.
NH4OH + HCl ⟶
Concept: undefined >> undefined
Reaction of an alcohol with a carboxylic acid in the presence of concentrated \[\ce{H2SO4}\] is termed as ______.
Concept: undefined >> undefined
