Advertisements
Advertisements
Classify the following amine as primary, secondary or tertiary:
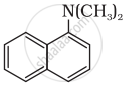
Concept: undefined >> undefined
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
Concept: undefined >> undefined
Advertisements
Write the IUPAC name of the following compound.

Concept: undefined >> undefined
Explain the observation, at the end of each period, there is a slight increase in the atomic radius of d-block elements.
Concept: undefined >> undefined
Write the expression of integrated rate equation for zero order reaction.
Concept: undefined >> undefined
What are Homoleptic complexes?
Concept: undefined >> undefined
Why chelate complexes are more stable than complexes with unidentate ligands?
Concept: undefined >> undefined
Name the element of 3d series which exhibits the largest number of oxidation states. Give reason.
Concept: undefined >> undefined
What are Heteroleptic complexes?
Concept: undefined >> undefined
Give reason for the following statements:
Scandium (Z = 21) is a transition element but Zn (Z = 30) is not.
Concept: undefined >> undefined
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
Concept: undefined >> undefined
Define transition metals.
Concept: undefined >> undefined
Among the following, which is the strongest base?
Concept: undefined >> undefined
Assertion (A) : Copper is a non-transition element.
Reason (R) : Copper has completely filled d-orbitals in its ground state.
Concept: undefined >> undefined
What is a chelate complex?
Concept: undefined >> undefined
Identify A and B in the following:

Concept: undefined >> undefined
The slope in the plot of [R] Vs. time for a zero-order reaction is ______.
Concept: undefined >> undefined
A voltaic cell is made by connecting two half cells represented by half equations below:
\[\ce{Sn^{2+}_{ (aq)} + 2e^- -> Sn_{(s)}}\], E0 = − 0.14 V
\[\ce{Fe^{3+}_{ (aq)} + e^- -> Fe^{2+}_{ (aq)}}\], E0 = + 0.77 V
Which statement is correct about this voltaic cell?
Concept: undefined >> undefined
The following experimental rate data were obtained for a reaction carried out at 25°C:
\[\ce{A_{(g)} + B_{(g)} -> C_{(g)} + A_{(g)}}\]
| Initial [A(g)]/mol dm−3 | Initial [B(g)]/mol dm−3 | Initial rate/mol dm−3s−1 |
| 3.0 × 10−2 | 2.0 × 10−2 | 1.89 × 10−4 |
| 3.0 × 10−2 | 4.0 × 10−2 | 1.89 × 10−4 |
| 6.0 × 10−2 | 4.0 × 10−2 | 7.56 × 10−4 |
What are the orders with respect to A(g) and B(g)?
Concept: undefined >> undefined
Assertion (A): For a zero-order reaction, the unit of rate constant and rate of reaction are same.
Reason (R): Rate of reaction for zero order reaction is independent of concentration of reactant.
Concept: undefined >> undefined
