Advertisements
Advertisements
The ether
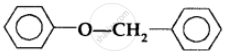
when treated with HI produces:
Concept: undefined >> undefined
In comparison to a 0.01 m solution of glucose, the depression in freezing point of a 0.01 m MgCl2 solution is ______.
Concept: undefined >> undefined
Advertisements
Which of the following statement is false?
Concept: undefined >> undefined
If molality of dilute solution is doubled, the value of molal depression constant (Kf) will be ______.
Concept: undefined >> undefined
Which has the highest freezing point?
Concept: undefined >> undefined
Ozone is a/an _______ molecule and the two O-O bond lengths in ozone are (i) _______-and (ii) _________.
Concept: undefined >> undefined
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Cryoscopic constant depends on nature of solvent.
Reason (R): Cryoscopic constant is a universal constant.
Select the most appropriate answer from the options given below:
Concept: undefined >> undefined
Which of the following statements is false?
Concept: undefined >> undefined
How does sprinkling of salt help in clearing the snow covered roads in hilly areas? Explain the phenomenon involved in the process.
Concept: undefined >> undefined
Assertion: When NaCl is added to water a depression in freezing point is observed.
Reason: The lowering of vapour pressure of a solution causes depression in the freezing point.
Concept: undefined >> undefined
What advantage do the fuel cells have over primary and secondary batteries?
Concept: undefined >> undefined
Match the items of Column I and Column II.
| Column I | Column II |
| (i) Lead storage battery | (a) maximum efficiency |
| (ii) Mercury cell | (b) prevented by galvanisation |
| (iii) Fuel cell | (c) gives steady potential |
| (iv) Rusting | (d) Pb is anode, PbO2 is cathode |
Concept: undefined >> undefined
Which of the following interface cannot be obtained?
Concept: undefined >> undefined
Method by which lyophobic sol can be protected?
Concept: undefined >> undefined
Which of the following colloids cannot be coagulated easily?
(i) Lyophobic colloids.
(ii) Irreversible colloids.
(iii) Reversible colloids.
(iv) Lyophilic colloids.
Concept: undefined >> undefined
What happens when gelatin is mixed with gold sol?
Concept: undefined >> undefined
Match the items given in Column I and Column II.
| Column I | Column II |
| (i) Protective colloid | (a) \[\ce{FeCl3 + NaOH}\] |
| (ii) Liquid-liquid colloid | (b) Lyophilic colloids |
| (iii) Positively charged colloid | (c) Emulsion |
| (iv) Negatively charged colloid | (d) \[\ce{FeCl3}\] + hot water |
Concept: undefined >> undefined
A number of elements are available in earth’s crust but most abundant elements are ______.
Concept: undefined >> undefined
Explain why ozone is thermodynamically less stable than oxygen.
Concept: undefined >> undefined
Coagulation of egg white on boiling is an example of denaturation of protein. Explain it in terms of structural changes.
Concept: undefined >> undefined
