Advertisements
Advertisements
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
p-Nitrobenzoic acid
Concept: undefined >> undefined
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
Phenylacetic acid
Concept: undefined >> undefined
Advertisements
How will you bring about the following conversion in not more than two steps?
Benazaldehyde to α-Hydroxyphenylacetic acid
Concept: undefined >> undefined
Complete the synthesis by giving missing starting material, reagent or product.
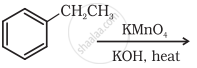
Concept: undefined >> undefined
Give the uses of freon 12.
Concept: undefined >> undefined
How the following conversion can be carried out?
Isopropyl alcohol to iodoform
Concept: undefined >> undefined
How the following conversion can be carried out?
Aniline to phenylisocyanide
Concept: undefined >> undefined
Write the structures of the monomers used for getting the following polymers
Melamine – formaldehyde polymer
Concept: undefined >> undefined
Write one difference between Multimolecular colloid and Macromolecular colloid
Concept: undefined >> undefined
Write one difference between Sol and Gel
Concept: undefined >> undefined
Write one difference between O/W emulsion and W/O emulsion
Concept: undefined >> undefined
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
Concept: undefined >> undefined
Write the product(s) in the following reactions
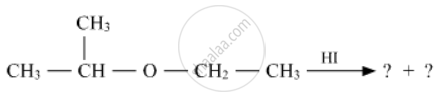
Concept: undefined >> undefined
Give simple chemical tests to distinguish between the following pairs of compounds :
Propanol and 2-methylpropan-2-ol
Concept: undefined >> undefined
Write one difference in Lyophobic sol and Lyophilic sol
Concept: undefined >> undefined
Why a solution of [Ni(H2O)6]2+ is green while a solution of [Ni(CN)4]2− is colourless? (At. no. of Ni = 28)
Concept: undefined >> undefined
Define the following term:
Colligative properties
Concept: undefined >> undefined
Write one difference between α-helix and β-pleated structures of proteins.
Concept: undefined >> undefined
Name the reagents used in the following reactions:

Concept: undefined >> undefined
Account for the following :
H2S has lower boiling point than H2O.
Concept: undefined >> undefined
