Please select a subject first
Advertisements
Advertisements
How is the variability in oxidation states of transition metals different from that of p-block elements?
Concept: undefined >> undefined
The oxidation state of Fe in [Fe(CO)5] is ______.
Concept: undefined >> undefined
Advertisements
The slope in the plot of ln[R] vs. time for a first order reaction is ______.
Concept: undefined >> undefined
Which of the following characteristics of transition metals is associated with their catalytic activity?
Concept: undefined >> undefined
Do the following conversions in not more than two steps:
\[\begin{array}{cc}
\ce{CH3CN to CH3 - C - CH3}\\
\phantom{...........}||\\
\phantom{...........}\ce{O}
\end{array}\]
Concept: undefined >> undefined
Do the following conversions in not more than two steps:

Concept: undefined >> undefined
Account for the following:
Sc3+ is colourless whereas Ti3+ is coloured in an aqueous solution.
Concept: undefined >> undefined
Account for the following:
Ce4+ is a strong oxidising agent.
Concept: undefined >> undefined
Assertion (A): Transition metals have high enthalpy of atomisation.
Reason (R): Greater number of unpaired electrons in transition metals results in weak metallic bonding.
Concept: undefined >> undefined
Which one among the following metals of the 3d series has the lowest melting point?
Concept: undefined >> undefined
The slope in the plot of `log ["R"]_0/(["R"])` Vs. time for a first-order reaction is ______.
Concept: undefined >> undefined
Which of the following transition metals shows +1 and +2 oxidation states?
Concept: undefined >> undefined
Assertion (A): Transition metals show their highest oxidation state with oxygen.
Reason (R): The ability of oxygen to form multiple bonds to metals.
Concept: undefined >> undefined
Do the following conversions in not more than two steps:
CH3COOH to CH3COCH3
Concept: undefined >> undefined
Do the following conversions in not more than two steps:
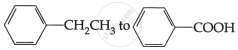
Concept: undefined >> undefined
Which property of transition metals enables them to behave as catalysts?
Concept: undefined >> undefined
Why is the C-O bond length in phenols less than that in methanol?
Concept: undefined >> undefined
Complete the following reaction and justify that it is a disproportionation reaction:
\[\ce{3MnO^{2-}4 + 4H^+ -> \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{} + 2H2O}\]
Concept: undefined >> undefined
The given graph shows the trends in melting points of transition metals:

Explain the reason why Cr has the highest melting point and manganese (Mn) has a lower melting point.
Concept: undefined >> undefined
Consider the following standard electrode potential values:
\[\ce{Fe^{3+}_{ (aq)} + e^- -> Fe^{2+}_{ (aq)}}\], E0 = +0.77 V
\[\ce{MnO^{-4}_{ (aq)} + 8H^+ + 5e^- -> Mn^{2+}_{ (aq)} + 4H2O_{(l)}}\], E0 = +1.51 V
What is the cell potential for the redox reaction?
Concept: undefined >> undefined
