Please select a subject first
Advertisements
Advertisements
Define the following modes of expressing the concentration of a solution. Which of these modes are independent of temperature and why?
(iii) w/V (mass by volume percentage)
Concept: undefined >> undefined
Define the following modes of expressing the concentration of a solution. Which of these modes are independent of temperature and why?
ppm. (parts per million)
Concept: undefined >> undefined
Advertisements
Define the following modes of expressing the concentration of a solution. Which of these modes are independent of temperature and why?
x (mole fraction)
Concept: undefined >> undefined
Define the following modes of expressing the concentration of a solution. Which of these modes are independent of temperature and why?
M (Molarity)
Concept: undefined >> undefined
Define the following modes of expressing the concentration of a solution. Which of these modes are independent of temperature and why?
m (Molality)
Concept: undefined >> undefined
Which cell will measure standard electrode potential of copper electrode?
Concept: undefined >> undefined
The difference between the electrode potentials of two electrodes when no current is drawn through the cell is called ______.
Concept: undefined >> undefined
Using the data given below find out the strongest reducing agent.
`"E"_("Cr"_2"O"_7^(2-)//"Cr"^(3+))^⊖` = 1.33 V `"E"_("Cl"_2//"Cl"^-) = 1.36` V
`"E"_("MnO"_4^-//"Mn"^(2+))` = 1.51 V `"E"_("Cr"^(3+)//"Cr")` = - 0.74 V
Concept: undefined >> undefined
Use the data given in below find out which of the following is the strongest oxidising agent.
`"E"_("Cr"_2"O"_7^(2-)//"Cr"^(3+))^⊖`= 1.33 V `"E"_("Cl"_2//"Cl"^-)^⊖` = 1.36 V
`"E"_("MnO"_4^-//"Mn"^(2+))^⊖` = 1.51 V `"E"_("Cr"^(3+)//"Cr")^⊖` = - 0.74 V
Concept: undefined >> undefined
The positive value of the standard electrode potential of Cu2+/Cu indicates that:
(i) this redox couple is a stronger reducing agent than the H+/H2 couple.
(ii) this redox couple is a stronger oxidising agent than H+/H2 .
(iii) Cu can displace H2 from acid.
(iv) Cu cannot displace H2 from acid.
Concept: undefined >> undefined
What does the negative sign in the expression `"E"^Θ ("Zn"^(2+))//("Zn")` = − 0.76 V mean?
Concept: undefined >> undefined
Value of standard electrode potential for the oxidation of \[\ce{Cl-}\] ions is more positive than that of water, even then in the electrolysis of aqueous sodium chloride, why is \[\ce{Cl-}\] oxidised at anode instead of water?
Concept: undefined >> undefined
Which reference electrode is used to measure the electrode potential of other electrodes?
Concept: undefined >> undefined
Assertion: Cu is less reactive than hydrogen.
Reason: `E_((Cu^(2+))/(Cu))^Θ` is negative.
Concept: undefined >> undefined
Consider the figure and answer the following question.
If cell ‘A’ has ECell = 0.5V and cell ‘B’ has ECell = 1.1V then what will be the reactions at anode and cathode?
Concept: undefined >> undefined
At high pressure the following reaction is zero order.
\[\ce{2NH3(g) ->[1130 K][Platinum catalyst] N2(g) + 3H2(g)}\]
Which of the following options are correct for this reaction?
(i) Rate of reaction = Rate constant.
(ii) Rate of the reaction depends on concentration of ammonia.
(iii) Rate of decomposition of ammonia will remain constant until ammonia disappears completely.
(iv) Further increase in pressure will change the rate of reaction.
Concept: undefined >> undefined
Which of the following graphs is correct for a zero order reaction?
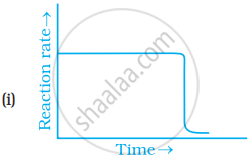

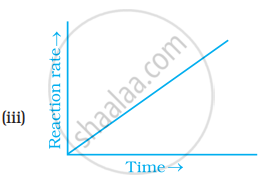

Concept: undefined >> undefined
Write the rate equation for the reaction `2A + B -> C` if the order of the reaction is zero.
Concept: undefined >> undefined
Derive an expression to calculate time required for completion of zero order reaction.
Concept: undefined >> undefined
For a zero order reaction will the molecularity be equal to zero? Explain.
Concept: undefined >> undefined
