Please select a subject first
Advertisements
Advertisements
Concept: undefined >> undefined
Concept: undefined >> undefined
Advertisements
Concept: undefined >> undefined
Concept: undefined >> undefined
Concept: undefined >> undefined
Concept: undefined >> undefined
Concept: undefined >> undefined
Concept: undefined >> undefined
Which of the following statement is true?
Concept: undefined >> undefined
Which of the following statements is false for a perfect conductor?
Concept: undefined >> undefined
Two a-particles have the ratio of their velocities as 3 : 2 on entering the field. If they move in different circular paths, then the ratio of the radii of their paths is ______.
Concept: undefined >> undefined
Let the magnetic field on earth be modelled by that of a point magnetic dipole at the centre of earth. The angle of dip at a point on the geographical equator is ______.
Concept: undefined >> undefined
Identify the wrong statement.
Concept: undefined >> undefined
Three Charges 2q, -q and -q lie at vertices of a triangle. The value of E and V at centroid of triangle will be ______.
Concept: undefined >> undefined
A solid spherical conductor has charge +Q and radius R. It is surrounded by a solid spherical shell with charge -Q, inner radius 2R, and outer radius 3R. Which of the following statements is true?
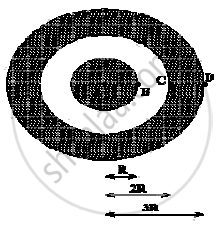
Concept: undefined >> undefined
Hydrogen spectrum consists of discrete bright lines in a dark background and it is specifically known as hydrogen emission spectrum. There is one more type of hydrogen spectrum that exists where we get dark lines on the bright background, it is known as absorption spectrum. Balmer found an empirical formula by the observation of a small part of this spectrum and it is represented by
`1/lambda = "R"(1/2^2 - 1/"n"^2)`, where n = 3, 4, 5,....
For Lyman series, the emission is from first state to nth state, for Paschen series, it is from third state to nth state, for Brackett series, it is from fourth state to nth state and for P fund series, it is from fifth state to nth state.
Number of spectral lines in hydrogen atom is ______.
Concept: undefined >> undefined
Hydrogen spectrum consists of discrete bright lines in a dark background and it is specifically known as hydrogen emission spectrum. There is one more type of hydrogen spectrum that exists where we get dark lines on the bright background, it is known as the absorption spectrum. Balmer found an empirical formula by the observation of a small part of this spectrum and it is represented by
`1/lambda = "R"(1/2^2 - 1/"n"^2)`, where n = 3, 4, 5,....
For Lyman series, the emission is from first state to nth state, for Paschen series, it is from third state to nth state, for Brackett series, it is from fourth state to nth state and for P fund series, it is from fifth state to nth state.
Which series of hydrogen spectrum corresponds to ultraviolet region?
Concept: undefined >> undefined
Hydrogen spectrum consists of discrete bright lines in a dark background and it is specifically known as hydrogen emission spectrum. There is one more type of hydrogen spectrum that exists where we get dark lines on the bright background, it is known as absorption spectrum. Balmer found an empirical formula by the observation of a small part of this spectrum and it is represented by
`1/lambda = "R"(1/2^2 - 1/"n"^2)`, where n = 3, 4, 5,....
For Lyman series, the emission is from first state to nth state, for Paschen series, it is from third state to nth state, for Brackett series, it is from fourth state to nth state and for P fund series, it is from fifth state to nth state.
Which of the following lines of the H-atom spectrum belongs to the Balmer series?
Concept: undefined >> undefined
Hydrogen spectrum consists of discrete bright lines in a dark background and it is specifically known as hydrogen emission spectrum. There is one more type of hydrogen spectrum that exists where we get dark lines on the bright background, it is known as absorption spectrum. Balmer found an empirical formula by the observation of a small part of this spectrum and it is represented by
`1/lambda = "R"(1/2^2 - 1/"n"^2)`, where n = 3, 4, 5,....
For Lyman series, the emission is from first state to nth state, for Paschen series, it is from third state to nth state, for Brackett series, it is from fourth state to nth state and for P fund series, it is from fifth state to nth state.
Rydberg constant is ______.
Concept: undefined >> undefined
What are eddy currents?
Concept: undefined >> undefined
