Advertisements
Advertisements
Choose the correct answer from the options given below:
A chloride which forms a precipitate that is soluble in excess of ammonium hydroxide is:
1) Calcium chloride
2) Ferrous chloride
3) Ferric chloride
4) Copper chloride
Concept: Action of Ammonium Hydroxide on Certain Salt Solutions
Identify the substance underlined, in the following case:
Cation that does not form a precipitate with ammonium hydroxide but forms one with sodium hydroxide.
Concept: Action of Sodium Hydroxide Solution on Certain Metallic Salt Solutions
State one relevant observation for given reactions:
Action of Sodium hydroxide solution on ferrous sulfate solution.
Concept: Action of Sodium Hydroxide Solution on Certain Metallic Salt Solutions
How will you distinguish between Ammonium hydroxide and Sodium hydroxide using copper sulphate solution?
Concept: Action of Ammonium Hydroxide on Certain Salt Solutions
Identify the salts P and Q from the observations given below:
On performing the flame test salt P produces a lilac coloured flame and its solution gives a white precipitate with silver nitrate solution, which is soluble in Ammonium hydroxide solution.
Concept: Action of Alkalis on Metal Oxides
_____________ (AgCl / PbCl2), a white precipitate is soluble in excess NH4OH
Concept: Action of Alkalis on Metal Oxides
Match the salts given in Column I with their method of preparation given in Column II.
| Column I | Column II | ||
| 1 | Pb(NO3)2 from PbO | A | Simple displacement |
| 2 | MgCl2 from Mg | B | Titration |
| 3 | FeCl3 from Fe | C | Neutralization |
| 4 | NaNO3 from NaOH | D | Precipitation |
| 5 | ZnCO3 from ZnSO4 | E | Combination |
Concept: Action of Alkalis on Metal Oxides
State your observations when ammonium hydroxide solution is added drop by drop and then in excess to each of the following solutions :
1) Copper sulphate solution
2) Zinc sulfate solution.
Concept: Action of Ammonium Hydroxide on Certain Salt Solutions
Identify the cations in the following case:
NaOH solution, when added to the Solution (A), gives a reddish brown precipitate
Concept: Action of Sodium Hydroxide Solution on Certain Metallic Salt Solutions
Choose the most appropriate answer from the following list of oxides which fit the description.
[SO2, SiO2, Al2O3, MgO, CO, Na2O]
A basic oxide.
Concept: Action of Alkalis on Certain Metals
Choose the most appropriate answer from the following list of oxides which fit the description
[SO2, SiO2, Al2O3, MgO, CO, Na2O]
An oxide which dissolves in water forming an acid
Concept: Action of Alkalis on Certain Metals
Choose the most appropriate answer from the following list of oxides which fit the description
[SO2, SiO2, Al2O3, MgO, CO, Na2O]
An amphoteric oxide
Concept: Action of Alkalis on Certain Metals
State your observation When excess sodium hydroxide is added to calcium nitrate solution
Concept: Action of Alkalis on Metal Oxides
Name the kind of particles present in sodium hydroxide solution.
Concept: Action of Sodium Hydroxide Solution on Certain Metallic Salt Solutions
State one relevant observation for the following.
Lead nitrate solution is treated with sodium hydroxide solution dropwise till it is excess.
Concept: Action of Alkalis on Certain Metals
A compound X (having the vinegar-like smell) when treated with ethanol in the presence of the acid Z, gives a compound Y which has a fruity smell.
The reaction is:
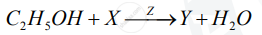
1) Identify Y and Z.
2) Write the structural formula of X.
3) Name the above reaction.
Concept: Action of Alkalis on Certain Metals
Give a chemical test to distinguish between the given pairs of chemicals:
Lead nitrate solution and Zinc nitrate solution
Concept: Action of Alkalis on Certain Metals
The hydroxide soluble in excess of ammonium hydroxide is ______.
Concept: Action of Alkalis on Metal Oxides
______ is a white precipitate that is soluble in excess of Ammonium hydroxide solution.
Concept: Action of Ammonium Hydroxide on Certain Salt Solutions
Identify the cation in the following case:
Ammonium hydroxide solution when added to Solution B gives a white precipitate which does not dissolve in excess of ammonium hydroxide solution.
Concept: Action of Ammonium Hydroxide on Certain Salt Solutions
