Advertisements
Advertisements
Which one of the following are the correct observations about acetic acid?
(A) It turns blue litmus red and smells like vinegar
(B) It turns blue litmus red and smells like burning sulphur
(C) It turns res litmus blue and smells like vinegar
(D) It turns red litmus blue and has a fruity smell
Concept: Ethanoic Acid
In a tabular form, differentiate between ethanol and ethanoic acid under the following heads:
(i) Physical state
(ii) Taste
(iii) NaHCO3 test
(iv) Ester test
Concept: Ethanol
Write a chemical reaction to show the dehydration of ethanol.
Concept: Ethanol
Acetic acid smells like:
(1) a banana
(2) vinegar
(3) an orange
(4) a lemon
Concept: Ethanoic Acid
Acetic acid solution turns:
(1) blue litmus red
(2) red litmus blue
(3) blue litmus colourless
(4) red litmus colourless
Concept: Ethanoic Acid
On adding NaHCO3 to acetic acid, a gas is evolved which turns lime water milky due to the formation of:
(1) Calcium bicarbonate
(2) Calcium hydroxide
(3) Calcium carbonate
(4) Calcium acetate
Concept: Ethanoic Acid
State two characteristic features of carbon which when put together give rise to a large number of carbon compounds.
Concept: Carbon: A Versatile Element
With a labelled diagram describe an activity to show the formation of an ester.
Concept: Ethanol
Distinguish between ionic and covalent compounds under the following properties:
(i) Strength of forces between constituent elements
(ii) Solubility of compounds in water
(iii) Electrical conduction in substances
Concept: The Covalent Bond
Explain the ways in which glucose is broken down in absence or shortage of oxygen.
Concept: Carbon: A Versatile Element
What is hydrocarbon? Give its one example.
Concept: Carbon: A Versatile Element
Give the structural difference between saturated and unsaturated hydrocarbons with two examples each.
Concept: Carbon: A Versatile Element
Name of the following compounds :

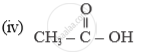
Concept: Carbon: A Versatile Element
List the conclusions you will draw while studying the following properties of ethanoic acid :
(a) Odour
(b) Solubility in water
(c) Effect on litmus paper
(d) Reaction with sodium hydrogen carbonate
Concept: Carbon: A Versatile Element
Answer the following question.
What is methane? Draw its electron dot structure. Name the type of bonds formed in this compound. Why are such compounds:
(i) poor conductors of electricity? and
(ii) have low melting and boiling points? What happens when this compound burns in oxygen?
Concept: The Covalent Bond
When sodium hydrogen carbonate is added to ethanoic acid, a gas evolves. Consider the following statements about the gas evolved.
- It turns lime water milky.
- It is evolved with brisk effervescence.
- It has a smell of burning sulfur.
- It is also a by-product of respiration.
Concept: Ethanoic Acid
Why is the conversion of ethanol to ethanoic acid considered an oxidation reaction? Write the oxidising agent used in the reaction involved.
Concept: Ethanol
Draw the structures for ethanol.
Concept: Ethanol
Assertion (A): In a homologous series of alcohols, the formula for the second member is C2H5OH and the third member is C3H7OH.
Reason (R): The difference between the molecular masses of the two consecutive members of a homologous series is 144.
Concept: Homologous Series
Distinguish between esterification and saponification reactions with the help of chemical equations for each.
Concept: Ethanoic Acid
