Definitions [2]
Define Allotrophy
When an element possess 2 or more different forms in the same state, they are called allotropes and the phenomenon is known as allotropy. Diamond and graphite are the 2 allotropes of carbon.
Define Allotrophy
When an element possess 2 or more different forms in the same state, they are called allotropes and the phenomenon is known as allotropy. Diamond and graphite are the 2 allotropes of carbon.
Key Points
General electronic configuration: ns² np¹.
| Element | Symbol | Electronic Configuration |
|---|---|---|
| Boron | B | [He] 2s² 2p¹ |
| Aluminium | Al | [Ne] 3s² 3p¹ |
| Gallium | Ga | [Ar] 3d¹⁰ 4s² 4p¹ |
| Indium | In | [Kr] 4d¹⁰ 5s² 5p¹ |
| Thallium | Tl | [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p¹ |
- All group 13 elements predominantly show a +3 oxidation state.
- Aluminium is the third most abundant element in Earth's crust (after oxygen and silicon).
- Boron is non-metallic; it does not react with non-oxidising acids but dissolves in strong oxidising acids like HNO₃ (conc.) and H₂SO₄ (conc.).
| Compound Name | Molecular Formula | Structural Feature | Important Uses |
|---|---|---|---|
| Borax | Na₂[B₄O₅(OH)₄]·8H₂O | 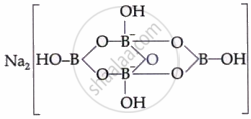 |
|
| Boric Acid | H₃BO₃ | 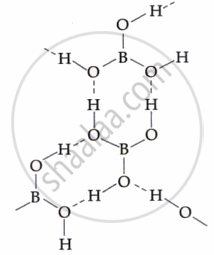 |
|
| Diborane | B₂H₆ | 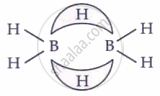 |
|
General electronic configuration: ns² np²
| Element | Symbol | Configuration |
|---|---|---|
| Carbon | C | [He] 2s² 2p² |
| Silicon | Si | [Ne] 3s² 3p² |
| Germanium | Ge | [Ar] 3d¹⁰ 4s² 4p² |
| Tin | Sn | [Kr] 4d¹⁰ 5s² 5p² |
| Lead | Pb | [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p² |
- Size & Energy Trends: Down the group, atomic and ionic radii increase, while ionisation energy decreases.
- Oxidation States: Show +4 and +2 states → +4 stable at top (C, Si), +2 becomes stable down the group (Sn, Pb) due to inert pair effect.
- Metallic Character: Increases down the group → C is non-metal, Si/Ge metalloids, Sn/Pb metals.
- Covalent Nature: Tendency to form covalent compounds decreases down the group as metallic character increases.
- Anomalous Behaviour of Carbon: Due to small size, high electronegativity, absence of d-orbitals, and high catenation.
- Catenation & Bonding: Carbon shows maximum catenation and strong covalent bonding, forming chains and rings.
Allotropes of Carbon:
- Crystalline: Diamond (hardest), Graphite (conducts electricity), Fullerene (spherical)
- Amorphous: Coke, Charcoal (adsorbent), Lampblack (~98–99% carbon)
General electronic configuration: ns² np³
| Element | Symbol | Configuration |
|---|---|---|
| Nitrogen | N | [He] 2s² 2p³ |
| Phosphorus | P | [Ne] 3s² 3p³ |
| Arsenic | As | [Ar] 3d¹⁰ 4s² 4p³ |
| Antimony | Sb | [Kr] 4d¹⁰ 5s² 5p³ |
| Bismuth | Bi | [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p³ |
- General Trends: Down the group → atomic size, density, boiling point increase, while ionisation enthalpy decreases.
- Melting Point Trend: Increases from N → As, then decreases from As → Bi.
- Electron Gain Enthalpy: Generally increases down the group (becomes more negative).
- Oxidation States: Show −3, +3, +5 → +5 stability decreases down the group due to the inert pair effect.
Important Compounds/Exceptions:
- PCl₅ exists as ionic solid → [PCl4]+[PCl6]−
- NF₃ is stable, while other trihalides are unstable
- Bond strength: PCl₃ > AsCl₃ > SbCl₃
Special Reactions:
- PH₃ is not spontaneously inflammable (unlike impure phosphine)
- With AgNO₃: forms Ag₃P (black ppt)
- With NH₃: forms (NH₄)₂PtCl₆ (yellow ppt)
Phosphorus Allotropes:
- White P: Reactive, glows in the dark (chemiluminescence), soluble in CS₂
- Red P: Stable, no glow
- Black P: Most stable, highest density, polymeric
General electronic configuration: ns² np³
| Element | Symbol | Configuration |
|---|---|---|
| Nitrogen | N | [He] 2s² 2p³ |
| Phosphorus | P | [Ne] 3s² 3p³ |
| Arsenic | As | [Ar] 3d¹⁰ 4s² 4p³ |
| Antimony | Sb | [Kr] 4d¹⁰ 5s² 5p³ |
| Bismuth | Bi | [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p³ |
- General Trends: Down the group → atomic size, density, boiling point increase, while ionisation enthalpy decreases.
- Melting Point Trend: Increases from N → As, then decreases from As → Bi.
- Electron Gain Enthalpy: Generally increases down the group (becomes more negative).
- Oxidation States: Show −3, +3, +5 → +5 stability decreases down the group due to the inert pair effect.
Important Compounds/Exceptions:
- PCl₅ exists as ionic solid → [PCl4]+[PCl6]−
- NF₃ is stable, while other trihalides are unstable
- Bond strength: PCl₃ > AsCl₃ > SbCl₃
Special Reactions:
- PH₃ is not spontaneously inflammable (unlike impure phosphine)
- With AgNO₃: forms Ag₃P (black ppt)
- With NH₃: forms (NH₄)₂PtCl₆ (yellow ppt)
Phosphorus Allotropes:
- White P: Reactive, glows in the dark (chemiluminescence), soluble in CS₂
- Red P: Stable, no glow
- Black P: Most stable, highest density, polymeric
General electronic configuration: ns² np⁴
| Element | Symbol | Configuration |
|---|---|---|
| Oxygen | O | [He] 2s² 2p⁴ |
| Sulphur | S | [Ne] 3s² 3p⁴ |
| Selenium | Se | [Ar] 3d¹⁰ 4s² 4p⁴ |
| Tellurium | Te | [Kr] 4d¹⁰ 5s² 5p⁴ |
| Polonium | Po | [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p⁴ |
- General Trends: Down the group → atomic size increases, while electronegativity and ionisation energy decrease.
- Oxidation States: Common states → −2, +2, +4, +6 (oxygen mainly −2; heavier elements show positive states).
- Chemical Reactivity: Decreases down the group → O > S > Se > Te > Po.
Hydrides Trend (H₂E):
- Bond angle decreases: H₂O > H₂S > H₂Se > H₂Te
- Thermal stability decreases down the group
- Acidic character increases: H₂O < H₂S < H₂Se < H₂Te
Sulphur Allotropes:
- Rhombic (α): Most stable, prepared from CS₂ solution
- Monoclinic (β): Formed by melting sulphur
Both consist of S₈ ring structure
Important Compounds & Reactions:
- H₂S: Poisonous, reducing agent
- SO₂: Acts as reducing + oxidising + bleaching agent
- O₃ (Ozone): Strong oxidising agent; converts KI → KIO₃/KIO₄
- O₂/O₃: Show allotropy
Sulphuric Acid (H₂SO₄):
- Dibasic acid
- Strong dehydrating agent
- Strong oxidising agent
- Low volatility & highly corrosive
General electronic configuration: ns² np⁴
| Element | Symbol | Configuration |
|---|---|---|
| Oxygen | O | [He] 2s² 2p⁴ |
| Sulphur | S | [Ne] 3s² 3p⁴ |
| Selenium | Se | [Ar] 3d¹⁰ 4s² 4p⁴ |
| Tellurium | Te | [Kr] 4d¹⁰ 5s² 5p⁴ |
| Polonium | Po | [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p⁴ |
- General Trends: Down the group → atomic size increases, while electronegativity and ionisation energy decrease.
- Oxidation States: Common states → −2, +2, +4, +6 (oxygen mainly −2; heavier elements show positive states).
- Chemical Reactivity: Decreases down the group → O > S > Se > Te > Po.
Hydrides Trend (H₂E):
- Bond angle decreases: H₂O > H₂S > H₂Se > H₂Te
- Thermal stability decreases down the group
- Acidic character increases: H₂O < H₂S < H₂Se < H₂Te
Sulphur Allotropes:
- Rhombic (α): Most stable, prepared from CS₂ solution
- Monoclinic (β): Formed by melting sulphur
Both consist of S₈ ring structure
Important Compounds & Reactions:
- H₂S: Poisonous, reducing agent
- SO₂: Acts as reducing + oxidising + bleaching agent
- O₃ (Ozone): Strong oxidising agent; converts KI → KIO₃/KIO₄
- O₂/O₃: Show allotropy
Sulphuric Acid (H₂SO₄):
- Dibasic acid
- Strong dehydrating agent
- Strong oxidising agent
- Low volatility & highly corrosive
Allotropes of Sulphur:
| Property | Rhombic (α) | Monoclinic (β) |
|---|---|---|
| Colour | Pale yellow | Bright yellow |
| Shape | Orthorhombic | Needle-shaped |
| Melting point | 385.8 K | 393 K |
| Density | 2.06 g/cm³ | 1.98 g/cm³ |
| Stability | Stable below 369 K | Stable above 369 K |
| Solubility | Insoluble in water, soluble in CS₂ | Soluble in CS₂ |
Allotropes of Group 16:
| Element | Allotropes |
|---|---|
| Oxygen | O₂, O₃ (ozone) |
| Sulfur | Rhombic, Monoclinic |
| Selenium | Red (non-metallic), Grey (metallic) |
| Tellurium | Crystalline, Amorphous |
| Polonium | α and β (both metallic) |
| Property | SO₂ (Sulphur dioxide) | H₂SO₄ (Sulphuric acid) |
|---|---|---|
| Preparation | FeS₂ + O₂ → SO₂ | Contact process |
| Physical | Colourless, pungent, poisonous gas | Colourless, dense, oily liquid |
| Nature | Reducing agent | Strong acid |
| Key reactions | With O₂ → SO₃ (V₂O₅) | Dehydrating agent |
| Special reaction | Decolourises KMnO₄ | Sugar → carbon |
| Other reactions | With NaOH → Na₂SO₃ | With salts → HCl |
| Uses | Bleaching, disinfectant | Fertilisers, industry |
| Name | Formula |
|---|---|
| Sulfurous acid | H₂SO₃ |
| Sulfuric acid | H₂SO₄ |
| Disulfuric (Pyrosulfuric) acid | H₂S₂O₇ |
| Peroxomonosulfuric acid | H₂SO₅ |
| Peroxodisulfuric acid | H₂S₂O₈ |
| Thiosulfuric acid | H₂S₂O₃ |
General electronic configuration: ns² np⁵
| Element | Symbol | Configuration |
|---|---|---|
| Fluorine | F | [He] 2s² 2p⁵ |
| Chlorine | Cl | [Ne] 3s² 3p⁵ |
| Bromine | Br | [Ar] 3d¹⁰ 4s² 4p⁵ |
| Iodine | I | [Kr] 4d¹⁰ 5s² 5p⁵ |
| Astatine | At | [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p⁵ |
Trends in Different Properties:
| Property | Trend |
|---|---|
| Enthalpy of Dissociation | Cl₂ > Br₂ > F₂ > I₂ |
| Dipole Moment | HF > HCl > HBr > HI |
| Ionic Character | HI < HBr < HCl < HF |
| Bond Length | HF < HCl < HBr < HI |
| Bond Strength | HI < HBr < HCl < HF |
| Thermal Stability | HI < HBr < HCl < HF |
| Reducing Power | HF < HCl < HBr < HI |
| Acid Strength | HF < HCl < HBr < HI |
General electronic configuration: ns² np⁵
| Element | Symbol | Configuration |
|---|---|---|
| Fluorine | F | [He] 2s² 2p⁵ |
| Chlorine | Cl | [Ne] 3s² 3p⁵ |
| Bromine | Br | [Ar] 3d¹⁰ 4s² 4p⁵ |
| Iodine | I | [Kr] 4d¹⁰ 5s² 5p⁵ |
| Astatine | At | [Xe] 4f¹⁴ 5d¹⁰ 6s² 6p⁵ |
Trends in Different Properties:
| Property | Trend |
|---|---|
| Enthalpy of Dissociation | Cl₂ > Br₂ > F₂ > I₂ |
| Dipole Moment | HF > HCl > HBr > HI |
| Ionic Character | HI < HBr < HCl < HF |
| Bond Length | HF < HCl < HBr < HI |
| Bond Strength | HI < HBr < HCl < HF |
| Thermal Stability | HI < HBr < HCl < HF |
| Reducing Power | HF < HCl < HBr < HI |
| Acid Strength | HF < HCl < HBr < HI |
| Oxidation state of X | Generic name | Oxoacids of fluorine | Oxoacids of chlorine | Oxoacids of bromine | Oxoacids of iodine |
|---|---|---|---|---|---|
| +1 | Hypohalous acid (HXO) | HOF | HOCl | HOBr | HOI |
| +3 | Halous acid (HXO₂) | – | HOClO | – | – |
| +5 | Halic acid (HXO₃) | – | HOClO₂ | HOBrO₂ | HOIO₂ |
| +7 | Perhalic acid (HXO₄) | – | HOClO₃ | HOBrO₃ | HOIO₃ |
Types of Interhalogen Compounds:
| Type | Examples |
|---|---|
| XX′ | ClF, BrF, ICl, IBr |
| XX′₃ | ClF₃, BrF₃, IF₃ |
| XX′₅ | ClF₅, BrF₅, IF₅ |
| XX′₇ | IF₇ |
Methods of Preparation:
| Method | Reactions |
|---|---|
| Direct combination | Cl₂ + F₂ → 2ClF Br₂ + 3F₂ → 2BrF₃ I₂ + 3Cl₂ → 2ICl₃ |
| Halogen + interhalogen | Br₂ + BrF₃ → 3BrF Br₂ + ClF₃ → 2BrF₃ + BrCl |
| Special (ICl) | I₂ + KClO₃ → ICl + KIO₃ |
Electronic Configurations:
- General electronic configuration: ns² np⁶ (He = 1s²)
- Elements: He, Ne, Ar, Kr, Xe, Rn
- Noble gases have very high ionisation enthalpies and positive electron gain enthalpies — so they generally do not form compounds.
- First noble gas compound prepared by Neil Bartlett: \[\mathrm{O}_{2}(\mathrm{g})+\mathrm{PtF}_{6}(\mathrm{g})\longrightarrow\mathrm{O}_{2}^{+}[\mathrm{PtF}_{6}]^{-}(\mathrm{s})\] — since the ionisation enthalpy of Xe ≈ that of O₂, Xe was reacted similarly.
Shapes and Hybridisation of Xenon Compounds:
| Molecule | Hybridisation | Geometry | Shape |
|---|---|---|---|
| XeO₃ | sp³ | Tetrahedral | Pyramidal |
| XeO₄ | sp³ | Tetrahedral | Tetrahedral |
| XeOF₂ | sp³d | Trigonal bipyramidal | T-shaped |
| XeOF₄ | sp³d² | Octahedral | Square pyramidal |
| XeO₂F₂ | sp³d | Trigonal bipyramidal | Distorted trigonal bipyramidal (see-saw) |
| XeO₃F₂ | sp³d | Trigonal bipyramidal | Trigonal bipyramidal |
| XeO₃F₄ | sp³d² | Octahedral | Octahedral |
Electronic Configurations:
- General electronic configuration: ns² np⁶ (He = 1s²)
- Elements: He, Ne, Ar, Kr, Xe, Rn
- Noble gases have very high ionisation enthalpies and positive electron gain enthalpies — so they generally do not form compounds.
- First noble gas compound prepared by Neil Bartlett: \[\mathrm{O}_{2}(\mathrm{g})+\mathrm{PtF}_{6}(\mathrm{g})\longrightarrow\mathrm{O}_{2}^{+}[\mathrm{PtF}_{6}]^{-}(\mathrm{s})\] — since the ionisation enthalpy of Xe ≈ that of O₂, Xe was reacted similarly.
Shapes and Hybridisation of Xenon Compounds:
| Molecule | Hybridisation | Geometry | Shape |
|---|---|---|---|
| XeO₃ | sp³ | Tetrahedral | Pyramidal |
| XeO₄ | sp³ | Tetrahedral | Tetrahedral |
| XeOF₂ | sp³d | Trigonal bipyramidal | T-shaped |
| XeOF₄ | sp³d² | Octahedral | Square pyramidal |
| XeO₂F₂ | sp³d | Trigonal bipyramidal | Distorted trigonal bipyramidal (see-saw) |
| XeO₃F₂ | sp³d | Trigonal bipyramidal | Trigonal bipyramidal |
| XeO₃F₄ | sp³d² | Octahedral | Octahedral |
Concepts [50]
- Introduction to p-block Elements
- Group 13 Elements - The Boron Family
- Boron
- Aluminum
- Important Compounds of Boron
- Some Important Compounds of Aluminium
- Group 14 Elements - The Carbon Family
- Important Trends and Anomalous Behaviour of Carbon
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Allotropes of Carbon > Diamond
- Some Important Compounds of Carbon and Silicon - Carbon Monoxide
- Some Important Compounds of Carbon and Silicon - Carbon Dioxide
- Some Important Compounds of Carbon and Silicon - Silicon Dioxide
- Some Important Compounds of Carbon and Silicon - Silicones
- Some Important Compounds of Carbon and Silicon - Silicates
- Some Important Compounds of Carbon and Silicon - Zeolites
- Group 15 Elements - The Nitrogen Family
- Group 15 Elements - The Nitrogen Family
- Dinitrogen
- Ammonia
- Oxides of Nitrogen
- Nitric Acid
- Phosphine
- Phosphorus Halides
- Phosphorus - Allotropic Forms
- Compounds of Phosphorus
- Oxoacids of Phosphorus
- Group 16 Elements - The Oxygen Family
- Group 16 Elements - The Oxygen Family
- Dioxygen
- Classification of Oxides
- Simple Oxides
- Ozone
- Sulphur - Allotropic Forms
- Compounds of Sulphur
- Sulphur Dioxide
- Oxoacids of Sulphur
- Sulphuric Acid
- Group 17 Elements - The Halogen Family
- Compounds of Halogens
- Group 17 Elements - The Halogen Family
- Chlorine
- Hydrogen Chloride
- Oxoacids of Halogens
- Interhalogen Compounds
- Trends in the Acidic Nature of Hydrogen Halides
- Group 18 Elements - The Noble gas Family
- Group 18 Elements - The Noble gas Family
