Definitions [6]
The valencies of all the atoms are satisfied by single bonds between them. Such carbon compounds are called saturated compounds.
The compounds of carbon having double or triple bonds between the carbon atoms are known as unsaturated carbon compounds.
Alkanes are hydrocarbons in which all the linkages between the carbon atoms are single covalent bonds.
Alkenes are unsaturated hydrocarbons containing at least one C=C double bond.
Compounds having the same molecular formula but different structural formula are known as Isomers and the phenomenon is known as Isomerism. (iso = same, meros = parts).
Alkynes are aliphatic unsaturated hydrocarbons containing at least one C≡C triple bond.
Key Points
- General formula: CₙH₂ₙ₊₂ (where n = number of carbon atoms)
- Suffix used for IUPAC naming: –ane
- e.g., Methane (CH₄), Ethane (C₂H₆), Propane (C₃H₈)
- Alkanes exhibit chain isomerism due to absence of any functional group and the possibility of more than one chain type for the same molecular formula
- e.g., C₅H₁₂ forms n-pentane, neo-pentane, and iso-pentane
1. Polarity:
- Alkanes are non-polar
- Insoluble in polar solvents (water)
- Soluble in non-polar solvents
2. Boiling point:
Increases with molecular mass
State at SATP:
- C₁–C₄ → gases
- C₅–C₁₇ → liquids
- C₁₈+ → solids
Boiling point order (increasing):
- Neo-pentane < iso-pentane < n-pentane
Trend:
- Straight-chain > branched-chain
- More branching → lower BP
3. Melting point:
- Increases with molecular mass
- Symmetrical molecules → higher MP
- Even number of C atoms → higher MP than odd
- Only intermolecular London forces are present
Alkanes are saturated and relatively inert — they undergo only substitution reactions at C–H bonds.
| Reaction | Conditions |
|---|---|
| Halogenation |
\[\ce{CH4 + Cl2 ->[hv][-HCl] CH3Cl ->[hv][-HCl] CH2Cl2 ->[hv][-HCl] CHCl3 ->[hv][-HCl] CCl4}\] |
| Combustion |
\[\ce{C_{n}H_{2n + 2} + \left(\frac{3n + 1}{2}\right)O_{2} ->[Complete][combustion] nCO2 + (n + 1)H2O}\] \[\ce{CH4(g) + 20_{2}(g) ->[Complete][combustion] CO2 + 2H2O}\] |
| Reforming / Aromatization |
\[\ce{n-hexane ->[V2O5][\underset{12-20 atm}{773K}] C6H6 + 4H2}\] |
| Pyrolysis / Cracking | 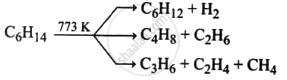 |
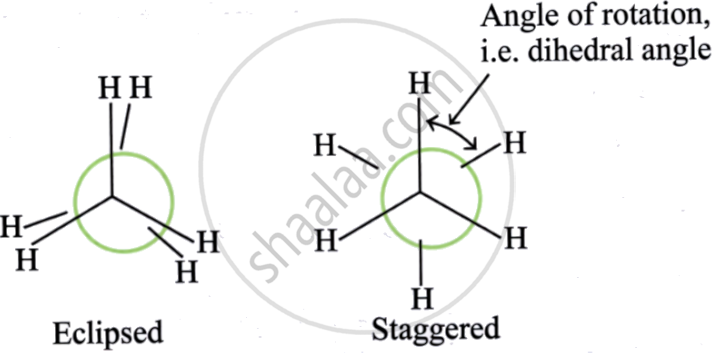
Conformations in Alkanes:
Conformation = spatial arrangement of atoms that can be interconverted by rotation about a C–C single bond. Also called conformers or rotamers.
For ethane, two extreme conformations exist:
| Conformation | Description | Energy | Stability |
|---|---|---|---|
| Eclipsed | Hydrogen atoms on adjacent carbons are as close as possible | Maximum energy | Highly unstable |
| Staggered | Hydrogen atoms are as far apart as possible | Minimum energy | Highly stable |
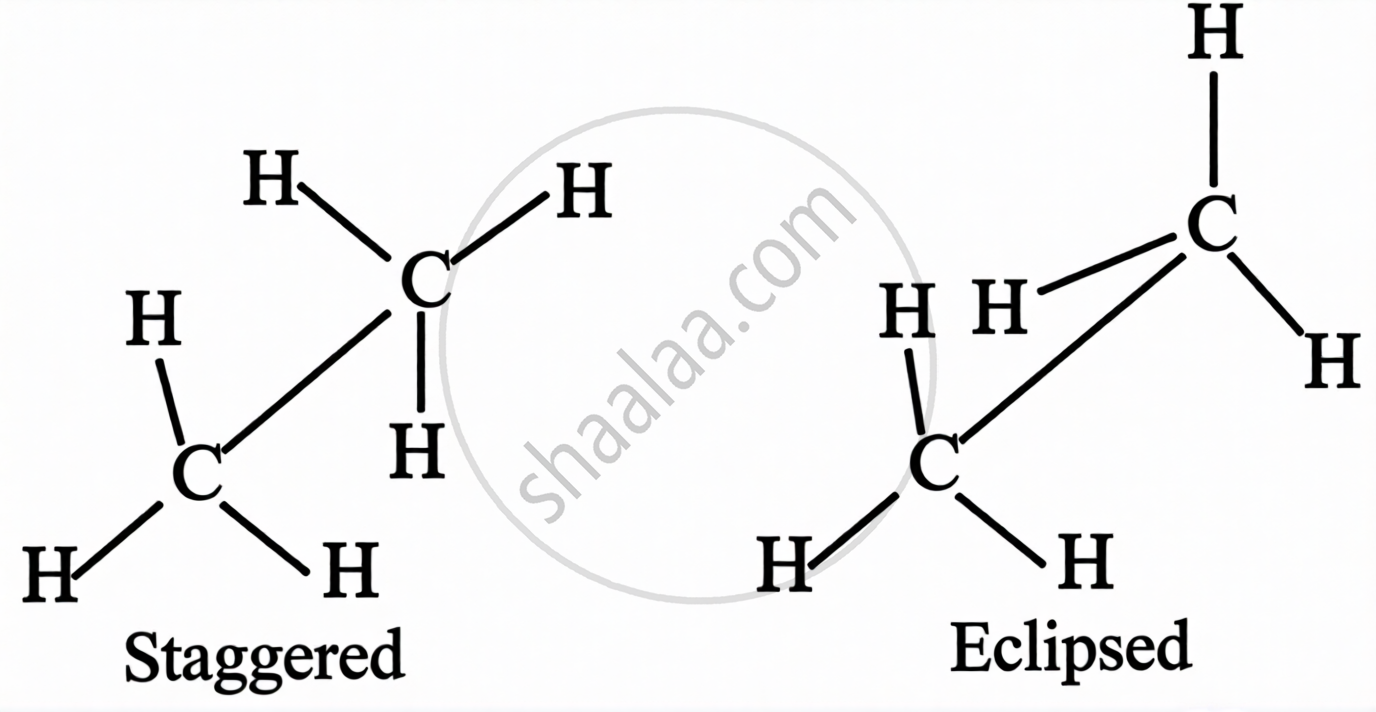
Sawhorse formula of ethane
- General formula: CₙH₂ₙ (where n = 2, 3, 4…)
- Suffix for IUPAC naming: –ene
- e.g., Ethene (CH₂=CH₂), Propene (CH₃–CH=CH₂)
- The double bond consists of one σ bond and one π bond
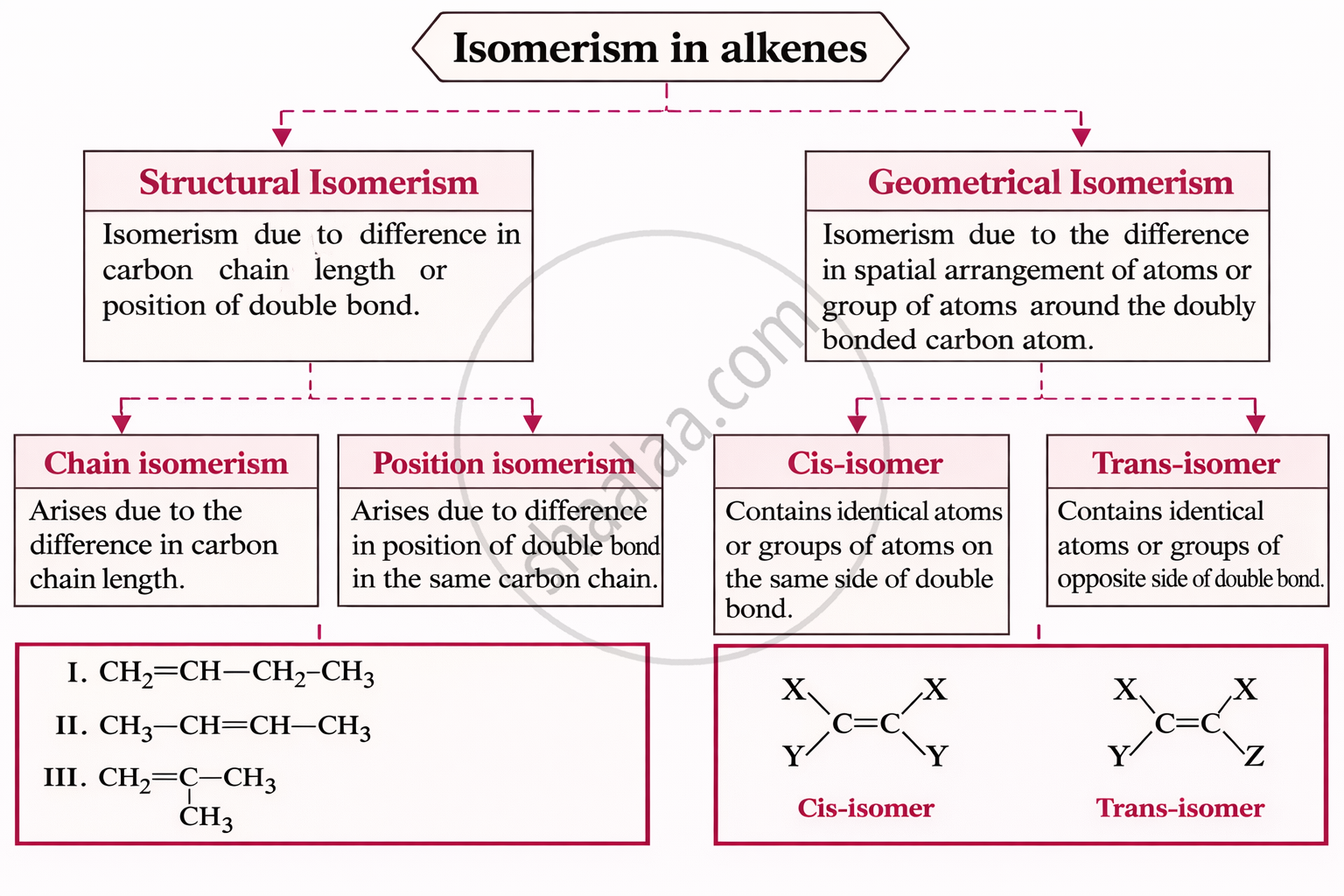
1. Structural Isomerism
Same molecular formula but different connectivity (bonding) of atoms.
Types:
- Chain isomerism: Different carbon skeleton (straight/branched)
- Position isomerism: Functional group at different positions
- Functional isomerism: Different functional groups (e.g., alcohol vs ether)
- Metamerism: Different alkyl groups on either side of same functional group
- Tautomerism: Dynamic equilibrium between two forms (keto ↔ enol)
2. Stereoisomerism
Same molecular formula and bonding but different spatial arrangement.
Types:
I. Geometrical isomerism:
- Due to restricted rotation (double bond)
- Forms cis (same side) and trans (opposite side)
II. Optical isomerism:
- Mirror image isomers (enantiomers)
- Show optical activity (rotate plane polarized light)
1. Solubility
- Alkenes are non-polar
- Insoluble in water
- Soluble in non-polar organic solvents (e.g., hexane, benzene)
2. Boiling Point (BP)
Increases with molecular mass
- More electrons → stronger London dispersion forces
Straight-chain > Branched-chain
- Straight chains have larger surface area → stronger intermolecular forces
Cis-alkenes > Trans-alkenes (usually)
- Cis is more polar → dipoles do not cancel → higher BP
Alkenes vs Alkanes (same number of carbons)
- Alkenes have slightly lower BP
Reason:
- π-bond leads to less effective electron distribution for dispersion forces
- Slightly weaker intermolecular attractions
3. Melting Point (MP)
Trans-alkenes > Cis-alkenes
- Trans is more symmetrical → packs better in crystal lattice → higher MP
Cis-alkenes
- Less symmetrical → poorer packing → lower MP
In trans-alkenes:
- Bond dipoles cancel → non-polar
- Leads to tighter packing in solid state
Alkenes undergo mainly electrophilic addition reactions due to the π bond (electron-rich site).
| Reaction | Example |
|---|---|
| Addition of hydrogen (Hydrogenation) |
\[\ce{H2C = CH2 ->[H2/Ni, Pt or Pd][523-573K] H3C - CH3}\] |
| Addition of halogen | \[\begin{array}{cc} \phantom{}\ce{H3CCH = CH2 + Cl — Cl ->[CCl4] CH3CH - CH2}\phantom{}\\ \phantom{....................................................}|\phantom{.........}|\phantom{}\\ \phantom{.....................................................}\ce{Cl}\phantom{.......}\ce{Cl}\phantom{} \end{array}\] |
| Addition of HX (Markovnikov's rule) | \[\begin{array}{cc} \phantom{..............................................................}\ce{Br}\phantom{}\\ \phantom{............................................................}|\phantom{}\\ \phantom{}\ce{\underset{(For unsymmetrical allkene-Markownikoff’s rule)}{H3CCH = CH2 + HBr} -> H3C - CH - CH3}\phantom{} \end{array}\] |
| Addition of HBr (Anti-Markovnikov / Kharasch effect) |
\[\begin{array}{cc} |
| Hydration (addition of H₂SO₄/H₂O) | \[\begin{array}{cc} \ce{O}\phantom{..}\\ ||\phantom{..}\\ \phantom{}\ce{CH2 = CH2 + H - O - S - O - H -> C2H5HSO4}\phantom{}\\ ||\phantom{..}\\ \ce{O}\phantom{..} \end{array}\] |
| Oxidation (KMnO₄/H⁺) | \[\begin{array}{cc} \phantom{..........................}\ce{O}\phantom{}\\ \phantom{..........................}||\\ \phantom{}\ce{H3C — CH = CH2 ->[{[O]}][KMnO4, {[H^{+}]}] H3C - C - OH + CO2 + H2O}\phantom{} \end{array}\] |
| Hydroxylation | \[\begin{array}{cc} \phantom{}\ce{H2C = CH2 + H2O + [O] ->[Dil.KMnO4][273K] CH2 - CH2}\\ \phantom{.....................................................}|\phantom{..........}|\phantom{}\\ \phantom{........................................................}\ce{OH}\phantom{.....}\ce{OH}\phantom{} \end{array}\] |
| Ozonolysis | \[\begin{array}{cc} \phantom{.....}\ce{H3C}\phantom{....................................}\ce{H3C}\phantom{............................}\\ \phantom{.....}\backslash\phantom{.........................................}\backslash\phantom{.....................}\\ \phantom{..........}\ce{C = CH2 + O3 ->[Zn/H2O] \phantom{.......}C = O + HCHO}\phantom{}\\ \phantom{......}/\phantom{..........................................}/\phantom{.....................}\\ \phantom{...............}\ce{H3C}\phantom{......................................}\ce{H3C}\phantom{......................................} \end{array}\] |
| Polymerisation | |
| Hydroboration-oxidation | \[\begin{array}{cc} \phantom{...............}\ce{H}\phantom{....}\ce{H}\phantom{.............................................}\ce{H}\phantom{....}\ce{H}\phantom{..........................}\ce{H}\phantom{....}\ce{H}\phantom{............................}\\ \phantom{.............}|\phantom{......}|\phantom{..............................................}|\phantom{......}|\phantom{............................}|\phantom{......}|\phantom{..........................}\\ \phantom{}\ce{6(H - C = C - H) + (BH3)2 ->[THF] 2(H - C - C)3 - B ->[H2O2][OH^Θ] H - C - C - H + B(OH)3}\phantom{}\\ \phantom{..........................................}|\phantom{......}|\phantom{............................}|\phantom{......}|\\ \phantom{..............................................}\ce{H}\phantom{.....}\ce{H}\phantom{..........................}\ce{H}\phantom{.....}\ce{OH}\phantom{.} \end{array}\] |
- General formula: CₙH₂ₙ₋₂
- Suffix for IUPAC naming: –yne
- e.g., Propyne (CH₃–C≡CH), Butyne (CH₃–CH₂–C≡CH)
- C₂H₂ is acetylene (common name); IUPAC name is ethyne
- The triple bond consists of one σ bond and two π bonds
Isomerism in Alkynes:
- Alkynes show position isomerism (type of structural isomerism)
- e.g., 1-Butyne and 2-Butyne
From calcium carbide:
By the action of water on calcium carbide, Cu2C2 or BaC2, acetylene is formed.
CaC2 + 2H2O → CH ≡ CH + Ca(OH)2
\[\ce{CaCO3 ->[][\underset{}{-CO2}] CaO->[2C + Heat][\underset{}{}] CaC2->[H2O][\underset{}{}] CH ≡ CH}\]
Dehalogenation of 1,1,2,2-tetrahaloalkanes:
On treatment with Zn, tetrahalides get dehalogenated to give alkynes.
\[\ce{R - CBr2 - CHBr2 + 2Zn ->[Δ] R - \underset{Alkyne}{C ≡ CH} + 2ZnBr2}\]
\[\ce{R - CBr2 - CBr2 - R + 2Zn ->[Δ] R - \underset{Alkyne}{C ≡ C} - R + 2ZnBr2}\]
By dehydrohalogenation of vic and gem dihalides:
Alkynes are prepared by dehydrohalogenation of vic. and gem. dihalides with alc. KOH + NaNH2 or KNH2.
\[\ce{R - \underset{β}{C}H2 - CHX2 ->[(i) NaNH2/Δ][(ii) H^{+}] R - C ≡ CH}\]
\[\begin{array}{cc}
\ce{X}\phantom{.......................}\\
\phantom{}|\phantom{.......................}\\
\phantom{}\ce{R - \underset{β}{C}H2 - C - R ->[NaNH2/Δ] R - C ≡ CH}\phantom{}\\
\phantom{}|\phantom{.......................}\\
\ce{X}\phantom{.......................}
\end{array}\]
Odour: Alkynes are generally odourless, but acetylene smells of garlic due to phosphine impurity
Boiling and melting points: Slightly higher than those of corresponding alkenes and alkanes with similar carbon atoms
| Order | BP/MP Comparison |
|---|---|
| BP of alkynes | Hex-1-yne > Pent-1-yne > But-1-yne > Propyne > Ethyne |
| MP of alkynes | Ethyne > Pent-1-yne > Propyne > But-1-yne > Hex-1-yne |
Reason for higher BP: Linear structure around the triple bond allows electrons to come closer together, resulting in greater London forces
Solubility: Alkynes are soluble in organic solvents like benzene, CCl₄, and ether
Density: Increases with increase in molecular size
Non-polar molecules → insoluble in water
| Reaction | Examples |
|---|---|
| Acidity of terminal alkynes |
\[\ce{HC ≡ CH + Na -> HC ≡ C^⊖Na^{+} + 2H2}\] |
| Addition of hydrogen (Hydrogenation) |
\[\ce{CH3C ≡ CH + 2H2 ->[Ni] CH3CH2CH3}\] |
| Addition of halogen | \[\begin{array}{cc} \phantom{..........................}\ce{Br}\phantom{...}\ce{Br}\phantom{}\\ \phantom{.........................}|\phantom{......}|\phantom{}\\ \phantom{}\ce{HC ≡ CH ->[2Br2][CCl4] H - C - C - H}\phantom{}\\ \phantom{.........................}|\phantom{......}|\phantom{}\\ \phantom{..........................}\ce{Br}\phantom{...}\ce{Br}\phantom{} \end{array}\] |
| Addition of HX |
\[\ce{HC ≡ CH + 2HBr -> CH3CHBr2}\] |
| Hydration (Hydration of alkynes) | \[\ce{HC ≡ CH + H2O ->[1\% HgSO4][40\% H2SO4] [\underset{Unstable}{CH2 = CHOH}] ⇌ \underset{\underset{compound}{Carbonyl}}{CH3CHO}}\] |
Cyclic, planar hydrocarbons with delocalised π electrons.
Benzenoids contain benzene ring; Non-benzenoids are aromatic without benzene ring.
Structure of Benzene:
- All 6 C atoms are sp² hybridised
- Unhybridised p-orbitals overlap laterally → delocalised π bonds
- Bond length = 139 pm (uniform, due to resonance)
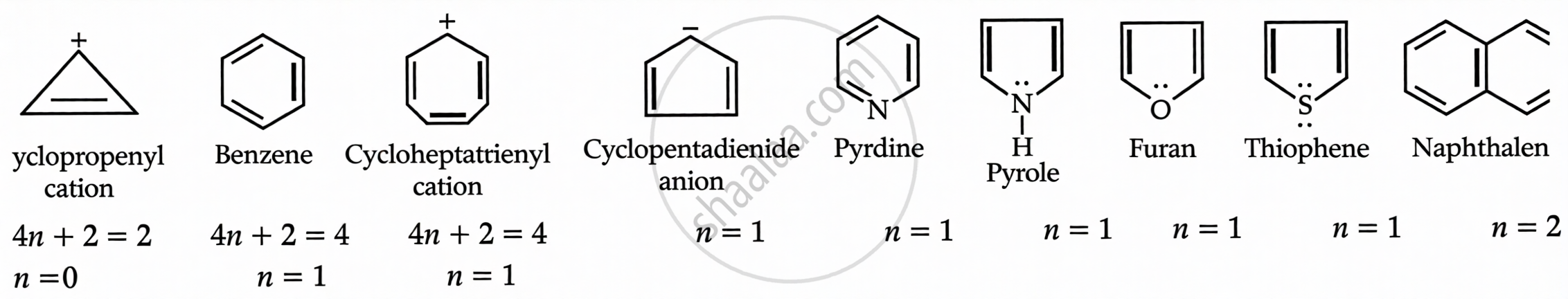
Huckel's Rule
- Cyclic and planar
- Each ring atom has a p-orbital
- Contains (4n + 2) π electrons (n = 0, 1, 2…)
Examples: Benzene, Naphthalene, Pyridine.
Physical Properties:
- Colourless liquid, sweet smell
- BP = 353 K, MP = 278.5 K
- Immiscible with water; burns with sooty flame
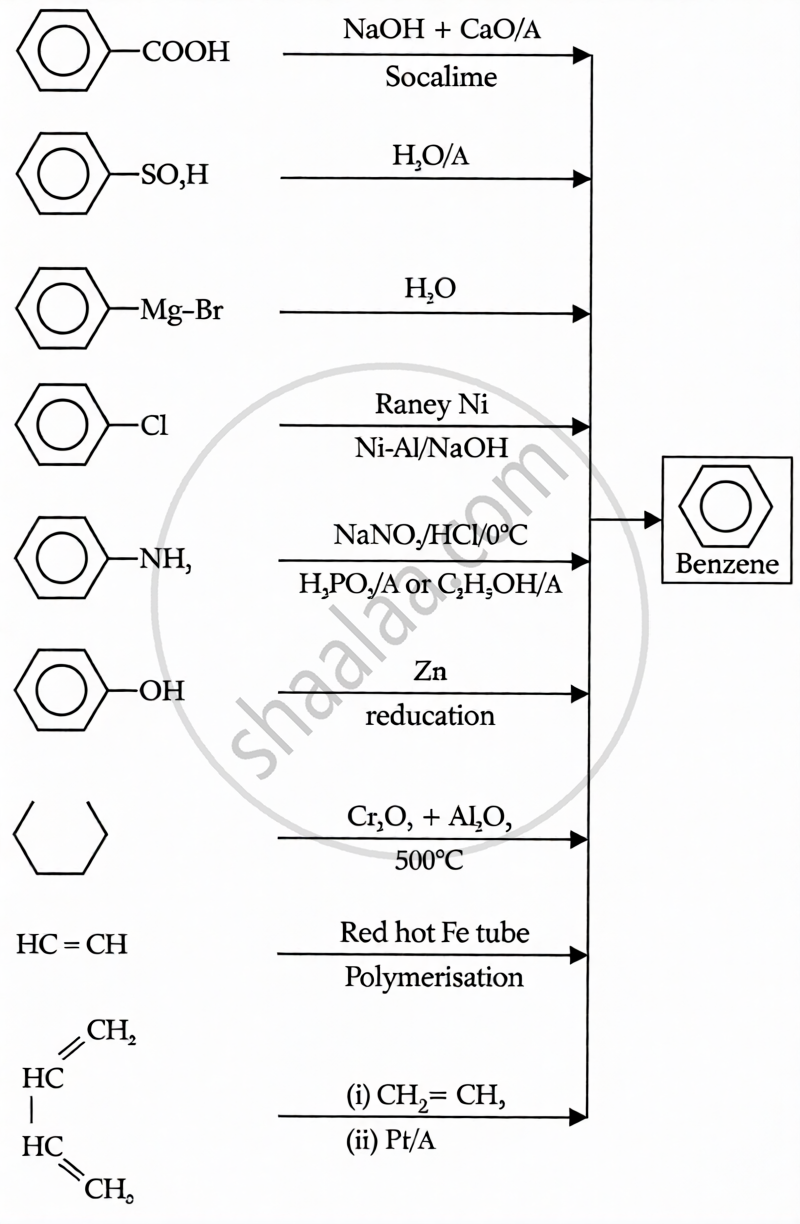
Preparation:
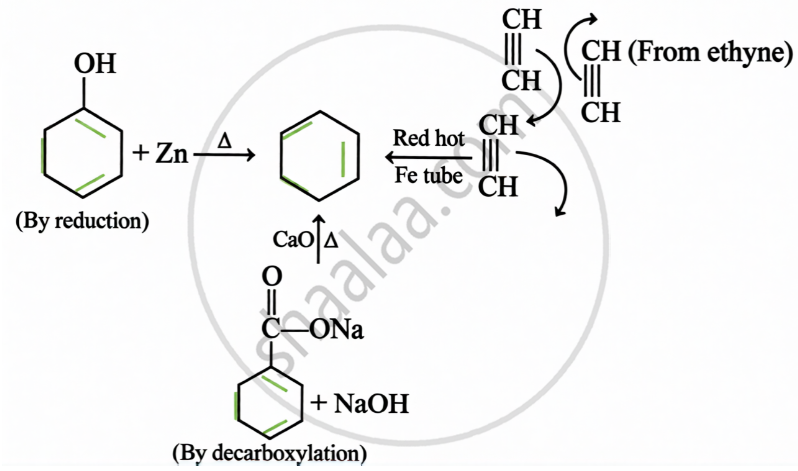
Electrophilic Substitution:
Aromatic compound are cyclic and planar, Aromaticity is due to extensive delocalisation of π-electrons in planar ringsystem.
Condition of Aromatic Nature: A compound will be aromatic if.
- Compound should be cyclic
- Compound should be planer
- Compound should be conjugated
It obey Hukel rule (4n + 2) = ne value of n will be 0, 1,3
- Aromatic hydrocarbons are non-polar molecules and are usually colourless liquids or solids with a characteristic aroma.
- Aromatic hydrocarbons are immiscible with water but are readily miscible with organic solvents.
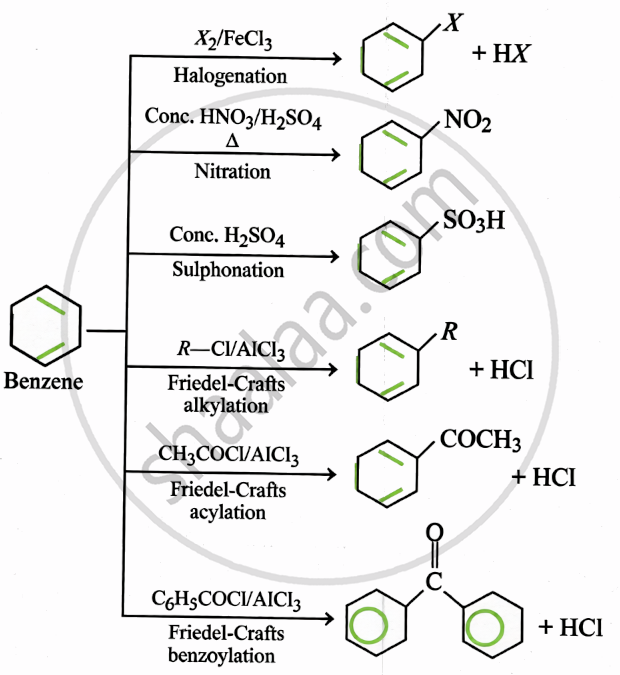
(i) Electrophilic substitution:

(ii) Addition reaction:

(iii) Friedel-Crafts reactions:
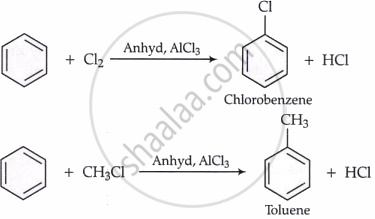
(iv) Friedel-Crafts acylation:
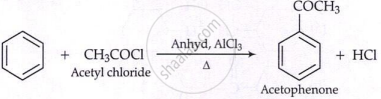
Aromatic hydrocarbons are electron-rich → give electrophilic substitution reactions (act as nucleophiles).
- Aromaticity is preserved in ESR because the double bond is not broken.
- E is the electrophile.

Electrophilic substitution reaction takes place in three steps:
- Generation of electrophile
- Formation of carbocation
- Removal of proton
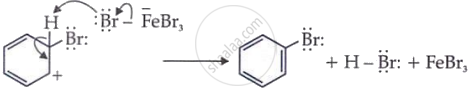
Step 1: Generation of Electrophile
In the first step of the electrophilic substitution reaction, the electrophile is generated with the help of a Lewis acid example H+, FeX3, or AICl3
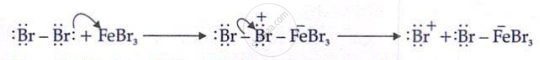
Step 2: Electrophile attack on benzene ring
The positive bromine ion attacks benzene to form an arenium ion.
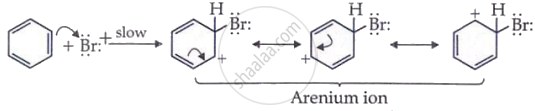
Step 3: Deprotonation from benzene ring
A proton is removed from the arenium ion to become bromobenzene.
- Benzene and polynuclear hydrocarbons (containing two or more fused benzene rings) are toxic and may show carcinogenic (cancer-causing) properties.
- Some polynuclear hydrocarbons like benzo[a]pyrene are strongly carcinogenic.
- Anthracene itself is NOT carcinogenic, but some of its derivatives may be.
- Therefore, compounds containing benzene rings should be handled with care.
Concepts [31]
- Classification of Hydrocarbons
- Alkanes
- Nomenclature and Isomerism
- Preparation of Alkanes from Unsaturated Hydrocarbons, Alkyl Halides and Carboxylic Acids
- Physical Properties of Alkanes
- Chemical Properties of Alkanes
- Conformations (Ethane)
- Alkenes
- Structure of Double Bond (Ethene)
- Nomenclature
- Isomerism
- Preparation of Alkenes from Alkynes, Alkyl Halides, Vicinal Dihalides and Alcohols by Acidic Dehydration
- Physical Properties of Alkenes
- Chemical Properties of Alkenes
- Alkynes
- Nomenclature and Isomerism
- Structure of Triple Bond
- Preparation of Alkynes
- Physical Properties of Alkynes
- Chemical Properties of Alkynes
- Aromatic Hydrocarbons
- Nomenclature and Isomerism
- Structure of Benzene
- Aromaticity (Huckel Rule)
- Preparation of Benzene
- Physical Properties of Aromatic Hydrocarbons
- Chemical Properties of Aromatic Hydrocarbons
- Electrophilic Substitution Reactions
- Concept of Reactivity in Electrophilic Substitution ESR
- Directive Influence of a Functional Group in Monosubstituted Benzene
- Carcinogenicity and Toxicity
