Definitions [2]
Compounds in which one or more hydrogen atom(s) of an alkane is (are) replaced by halogen.
General formula: R–X.
Compounds in which one or more hydrogen atom(s) directly bonded to an aromatic ring is (are) replaced by halogen.
General formula: Ar–X.
Key Points
Aryl halides are less reactive than alkyl halides in nucleophilic substitution. Due to resonance effect, lone pair on halogen is delocalized into benzene ring.
This gives partial double bond character to C–X bond → bond becomes shorter & stronger.
Strong electron-withdrawing groups (EWGs) like –NO₂ increase reactivity.
EWGs must be at ortho or para positions for effective substitution.
Example reaction:
p-chloronitrobenzene + OH⁻ → p-nitrophenol (Cl replaced by OH).
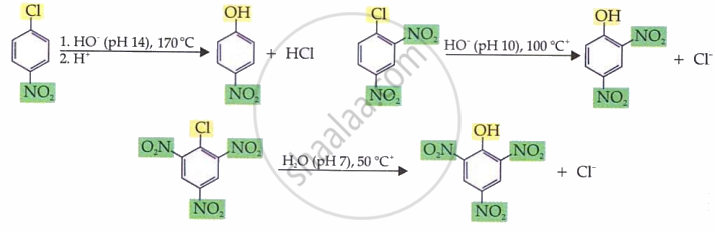
Mechanism is SNAr (Addition–Elimination).
- Step 1: Nucleophile attacks carbon bearing halogen → forms intermediate.
- Step 2: Leaving group (Cl⁻) departs → aromaticity restored.
- Reactivity order:
More –NO₂ groups = higher reactivity
(Tri-NO₂ > Di-NO₂ > Mono-NO₂ > no EWG)
Important Questions [8]
- Which of the Following Carbocations is Least Stable?
- What is Grignard reagent?
- Why does p-nitrochlorobenzene undergo displacement reactions readily with attack of nucleophilic HOΘ ion?
- Explain Only Reaction Mechanism for Alkaline Hydrolysis of Tert-butylbromide
- Write a Note on Friedel Craft’S Acylation.
- How is chlorobenzene converted into diphenyl/biphenyl?
- Draw structure of DDT.
- Write the environmental effects of DDT.
