Definitions [11]
Bifunctional organic compounds containing a carboxylic and an amino group either at the same carbon atom or at nearby carbon atoms are called amino acids.
Define the term Protein.
Chemically proteins are polyamides which are high molecular weight polymers of the monomer units, i.e., α-amino acids. OR It can also be defined as proteins are the biopolymers of a large number of α-amino acids and they are naturally occurring polymeric nitrogenous organic compounds containing 16% nitrogen and peptide linkages (-CO-NH-)
Define enzymes.
Enzymes are biological catalysts that speed up chemical reactions in living cells without being consumed in the process.
Proteins are complex polyamides formed from amino acids. They are essential for the proper growth and maintenance of the body. They have many peptide (-CO–NH )bonds.
Chemically, proteins are polyamides, which are high molecular weight polymers of the monomer units called \[\alpha\]-amino acids.
Define α-amino acids.
α-Amino acids are carboxylic acids having an amino (–NH2) group bonded to the α-carbon, that is, the carbon next to the carboxyl (–COOH) group.
Define peptide bond.
The bond that connects α-amino acids to each other is called a peptide bond.
Define Zwitter ion
An ∝-amino acid molecule contains both acidic carboxyl (-COOH) group as well as basic amino (-NH2) group. Proton transfer from acidic group to basic group of amino acid forms a salt, which is a dipolar ion called zwitter ion.
Define the following term:
Essential amino acids
Amino acids that cannot be synthesised in the human body and must be obtained through diet are known as essential amino acids.
Define the following as related to proteins:
Peptide linkage
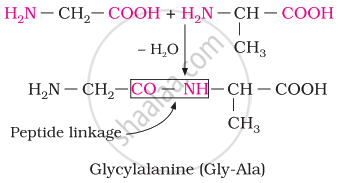
Chemically, peptide linkage is an amide formed between the –COOH group and –NH2 group. The reaction between two molecules of similar or different amino acids proceeds through the combination of the amino group of one molecule with the carboxyl group of the other. This results in the elimination of a water molecule and the formation of a peptide bond –CO–NH–. The product of the reaction is called a dipeptide because it is made up of two amino acids.
For example, when the carboxyl group of glycine combines with the amino group of alanine, we get a dipeptide, glycylalanine.
A colloidal solution of protein which works as a biological catalyst is known as an enzyme.
Key Points
- Proteins are polymers of amino acids (polypeptides) in which amino acids are linked by peptide bonds.
- There are 20 types of amino acids, so proteins are heteropolymers (not homopolymers).
- Amino acids are of two types: essential (must be obtained from diet) and non-essential (can be synthesised in the body).
- Proteins are high molecular weight biomolecules (polyamides) made of α-amino acids with a general structure R-CH(NH₂)-COOH.
- Proteins perform various functions such as enzymatic activity, transport, hormonal regulation, immunity, and sensory reception.
- Proteins are of two main types: fibrous proteins (insoluble, structural, e.g., keratin) and globular proteins (soluble, functional, e.g., enzymes, insulin).
- Collagen is the most abundant protein in animals, while RuBisCO is the most abundant enzyme in the biosphere.
α-Amino acids: Carboxylic acids where α-hydrogen is replaced by the –NH₂ group
General structure: \[ \mathrm{R} - \underset{\underset{\displaystyle \mathrm{NH}_2}{|}}{\overset{\overset{\displaystyle \alpha}{\displaystyle \mathrm{CH}}}{}} - \mathrm{COOH}\]
(where R = H or alkyl group at α-carbon)
Essential amino acids (not synthesised in the body; must be taken in food):
- Leucine
- Isoleucine
- Lysine
- Methionine
- Phenylalanine
- Threonine
- Tryptophan
- Valine
Non-essential amino acids (synthesised in the body):
- Alanine
- Asparagine
- Aspartic acid
- Cysteine
- Glutamic acid
- Glutamine
- Glycine
- Proline
- Serine
- Tyrosine
Semi-essential (50% body + 50% food):
- Arginine
- Histidine
Glycine is the only optically inactive amino acid (no chiral centre; R = H)
Isoelectric Point (pI):
- pH at which an amino acid does not migrate in an electric field → exists as a zwitterion (+NH₃–CH(R)–COO⁻)
- At pI, the concentration of zwitterions is maximum; anionic and cationic forms are equal
- At pI, an amino acid has the least solubility in water (used in separation by isoelectric precipitation)
- For neutral amino acid: \[pI=\frac{1}{2}(pk_{a_1}+pk_{a_2})\]
- Proteins are made up of α-amino acids linked together in a chain.
- A peptide bond (–CO–NH–) is formed between the carboxyl group of one amino acid and the amino group of another with the removal of water (condensation).
- Two amino acids form a dipeptide, three form a tripeptide, and many form polypeptides.
- Proteins are long polypeptide chains containing more than 100 amino acid residues.
- The ends of a protein chain are different:
The N-terminal has a free amino group, and the C-terminal has a free carboxyl group. - A peptide bond is similar to a secondary amide linkage in organic chemistry.
| Level | Description | Stabilising Forces |
|---|---|---|
| Primary | Linear sequence of amino acids linked by peptide bonds | Peptide bonds |
| Secondary | α-helix (right-handed coil, –NH of one AA H-bonded to C=O of 4th residue) OR β-pleated sheet (when R group is small) | Hydrogen bonds |
| Tertiary | Further folding of secondary structure; 3D shape | Hydrophobic interactions, H-bonds, disulphide bonds, van der Waals, ionic interactions |
| Quaternary | Two or more polypeptide chains arranged spatially | All forces present in tertiary structure |
- Enzymes are biological catalysts, mostly proteins, that increase the rate of biochemical reactions without being consumed.
- Some enzymes are ribozymes, which are RNA molecules that act like enzymes.
- Enzymes have primary, secondary, and tertiary structures, and their 3D structure determines their specificity and function.
- Each enzyme has a specific active site where the substrate binds to form an enzyme–substrate complex.
- Enzymes are highly specific and lower the activation energy of reactions.
- Enzyme activity is affected by temperature and pH; most enzymes are denatured at high temperatures, while thermophilic enzymes remain stable at 80–90°C.
- Examples of enzymes include amylase (starch → glucose), pepsin (proteins → amino acids), lactase (lactose → glucose + galactose), and maltase (maltose → glucose).
Mechanism of Enzyme Action (Lock and Key model):
- Enzyme (E) binds to substrate (S) → ES complex (E + S → ES)
- Product formation: ES → EP
- Product released: EP → E + P (enzyme regenerated)
- Enzymes work best at 298 K to 313 K (25°C to 40°C) — optimum temperature
- Activity decreases with temperature increase or decrease beyond optimum range; stops at ~273 K
Hormones are chemical substances produced by ductless (endocrine) glands; released into the bloodstream to regulate organ functions
Classified into: Steroid hormones, Amine hormones, Peptide hormones
| Type | Example | Gland | Function |
|---|---|---|---|
| Steroid | Testosterone | Testes | Development of male sex organs |
| Steroid | Oestrogen & Progesterone | Ovary | Female sex organ development; pregnancy |
| Steroid | Cortisone | Adrenal cortex | Regulates water and mineral salt metabolism |
| Amine | Adrenaline | Adrenal medulla | Increases blood pressure and pulse rate |
| Amine | Thyroxine | Thyroid | Stimulates oxidative metabolism; growth |
| Peptide | Oxytocin | Posterior pituitary | Contraction of uterus during childbirth |
| Peptide | Vasopressin | Posterior pituitary | Controls water reabsorption in kidneys |
| Peptide | Insulin | Pancreas (posterior pituitary) | Controls blood glucose (lowers) |
| Peptide | Glucagon | Pancreas | Increases blood glucose level |
Important Questions [18]
- Define peptide bond.
- Write the Correct Reaction for Formation of Peptide Bond Between Amino Acids
- What Are Amino Acids?
- How is Peptide Linkage Formed?
- How Are Proteins Classified on the Basis of Molecular Shapes?
- What is peptide linkage?
- How is tripeptide formed?
- Discuss the Optical Activity of Lactic Acid.
- Explain formation of peptide linkage in protein with an example.
- Write a classification of proteins with an example.
- Define Enzymes
- What are enzymes?
- Mention 'Two' Functions of Lipids
- Define Complex Lipids
- What Are Hormones?
- Write the Structure of Simple Triglycerides.
- The Secondary Structure of Protein is Determined by
- State the Function of Insulin.
