Advertisements
Advertisements
Question
Write the mechanism of the following reaction:
\[\ce{2CH3CH2OH ->[H^+][413 K] CH3-CH2-O-CH2-CH3 + H2O}\]
Advertisements
Solution
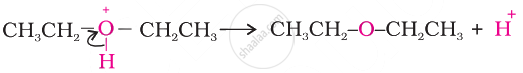
I.
\[\begin{array}{cc}
\phantom{.....................................}\ce{H}\\
\ce{CH3-CH2-\overset{\bullet\bullet}{\underset{\bullet\bullet}{O}}-H + H^+ -> CH3-CH2-\overset{+\bullet\bullet\phantom{...}}{\underset{\bullet\bullet}{O}}-H}
\end{array}\]
II.

III.
APPEARS IN
RELATED QUESTIONS
Write the reactions of Williamson synthesis of 2-ethoxy-3-methylpentane starting from ethanol and 3-methylpentan-2-ol.
Explain the following with an example.
Williamson ether synthesis
Dehydration of alcohol to ethers is catalysed by:
Explain why low molecular mass alcohols are soluble in water.
The Williamson ether synthesis produces ethers by reacting an
Name the suitable alcohol and reagent, from which 2-Chloro-2-methyl propane can be prepared.
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride->[i)CH3MgBr][ii)H3O+]X ->[acidk2crp3]A}\]
Identify the product (s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Give the structure and IUPAC name of the metamers of 2-methoxy propane.
