Advertisements
Advertisements
प्रश्न
Write the mechanism of the following reaction:
\[\ce{2CH3CH2OH ->[H^+][413 K] CH3-CH2-O-CH2-CH3 + H2O}\]
Advertisements
उत्तर
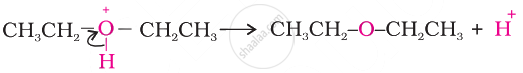
I.
\[\begin{array}{cc}
\phantom{.....................................}\ce{H}\\
\ce{CH3-CH2-\overset{\bullet\bullet}{\underset{\bullet\bullet}{O}}-H + H^+ -> CH3-CH2-\overset{+\bullet\bullet\phantom{...}}{\underset{\bullet\bullet}{O}}-H}
\end{array}\]
II.

III.
APPEARS IN
संबंधित प्रश्न
How do you convert the following :
Phenol to anisole
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
Explain the following with an example.
Williamson ether synthesis
Which of the following cannot be made by using Williamson Synthesis:
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Identify the product (s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Write the names of reagents and equations for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Give the structure and IUPAC name of the metamers of 2-methoxy propane.
