Advertisements
Advertisements
Question
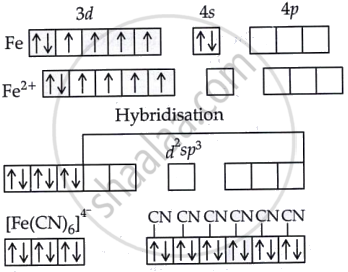
Write the hybridisation and magnetic behaviour of [Fe(CN)6]4−.
Very Short Answer
Advertisements
Solution
The inner orbital complex is formed by the d2sp3 hybridization. There is no magnetic moment because the complex is diamagnetic due to the absence of unpaired electrons.
shaalaa.com
Is there an error in this question or solution?
2024-2025 (March) Delhi Set 1
