Advertisements
Advertisements
Question
Write the answer to the questions with reference to the periodic table.

- What do (A), (B), (C), and (D) represent? [1]
- Elements are arranged in increasing order of their ______. [1]
- Write the electronic configuration of the first four elements in Group I. [1]
- What change in atomic radius is observed in moving from left to right in a Period? [1]
- Give a reason for (d). [1]
Advertisements
Solution
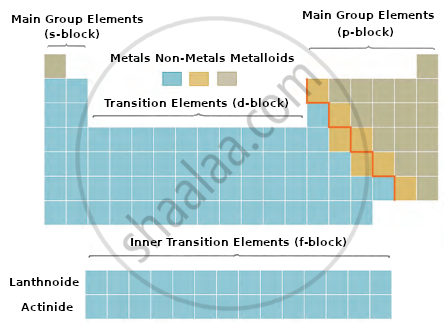
(A) represents s-block elements.
(B) represents p-block elements.
(C) represents transition elements (d-block).
(D) represents inner transition elements, i.e., (f-block).- Elements are arranged in increasing order of their atomic numbers.
- The electronic configuration for elements is as follows:
Elements Electronic configuration H 1 Li 2, 1 Na 2, 8, 1 K 2, 8, 8, 1 - Atomic radius goes on decreasing while going from left to right within a period.
-
The positive charge on the nucleus grows by one unit at a time as the atomic number rises sequentially while moving from left to right over time. However, the same outermost shell receives the additional electron as well. The electrons are more firmly drawn into the nucleus due to the higher nuclear charge, which reduces the size of the atom.
APPEARS IN
RELATED QUESTIONS
Write the name from the description.
The period with electrons in the shells K, L, and M.
Write the name from the description.
The family of metals having valency one.
Write the names from the description.
The metalloids in the second and third periods.
Match column A with column B.
|
Column A |
Column B |
|
(a) Element short by 1 electron in octet |
(i) Transition elements |
|
(b) Highly reactive metals |
(ii) Noble gases |
|
(c) Non-reactive elements |
(iii) Alkali metals |
|
(d) Elements of groups 3 to 12 |
(iv) Alkaline earth metals |
|
(e) Radioactive elements |
(v) Halogens |
|
(f) Elements with 2 electrons in the outermost orbit |
(vi)Actinides |
Write a short note on the following type of element –
transition elements
Write a short note on the following type of element –
inner transition elements
Write a short note on the following type of element –
normal elements
Name or state following with reference to the element of the first three periods of the periodic table.
The periods which contain the inner transition elements.
State whether true or false. If false, correct the statement.
Group 17 elements are named as Halogens.
Statement: Elements in a group generally possess similar properties but elements along a period have different properties.
Reason: The difference in electronic configuration makes the element differ in their chemical properties along a period.
d-block elements are otherwise known as ______.
If the electronic configuration of an element is 1s2 2s2 2p6 3s2 3p1’then it will
occupy ______ block of the periodic table.
f-block elements are also known as ______.
What are the s-block elements?
What are the p-block elements
Group 3-12 elements in the modern periodic table are called d block elements.
Answer the following questions related to the modern periodic table.
Name the blocks with the groups that they consist of.
Answer the following questions related to the modern periodic table.
Which block elements are known as transition elements?
Answer the following questions related to the modern periodic table.
How are elements placed around the zig-zag line in the p-block of the periodic table?
