Advertisements
Advertisements
Question
Which of the following compounds can be classified as aryl halides?
(i) \[\ce{p-ClC6H4CH2CH(CH3)2}\]
(ii) \[\ce{p-CH3CHCl(C6H4)CH2CH3}\]
(iii) \[\ce{o-BrH2C - C6H4CH(CH3)CH2CH3}\]
(iv) \[\ce{C6H5 - Cl}\]
Advertisements
Solution
(i) \[\ce{p-ClC6H4CH2CH(CH3)2}\]
(iv) \[\ce{C6H5 - Cl}\]
Explanation:
(i) 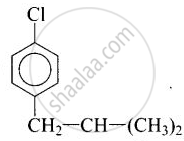
(ii) 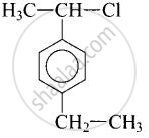
(iii) 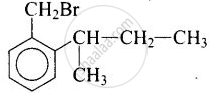
(iv) 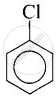
In compound (i) and compound (iv), halogen atom is directly bonded to aromatic ring therefore these compounds are classified as aryl halides.
APPEARS IN
RELATED QUESTIONS
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)2CHCH(Cl)CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH2CH(CH3)CH(C2H5)Cl
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(C2H5)2CH2Br
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH = C(Cl)CH2CH(CH3)2
When two halogen atoms are attached to the same carbon atom then it is:
Which is the correct IUPAC name for \[\begin{array}{cc}
\ce{CH3 - CH - CH2 - Br}\\
\phantom{}|\phantom{.......}\\
\phantom{}\ce{C2H5}\phantom{....}
\end{array}\]?
Which alkyl halide has maximum density
Two isomers (A) and (B) with molar mass 184 g/mol and elemental composition C 52.2%; H 4.9% and Br 42.9% gave benzoic acid and p-bromobenzoic acid, respectively on oxidation with KMnO4. Isomer ‘A’ is optically active and gives a pale yellow precipitate when warmed with alcoholic AgNO3. Isomer ‘A’ and ‘B’ are, respectively:
Which of the following belongs to the class of vinyl halides?
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3C(C2H5 )2CH2Br
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
IUPAC name of 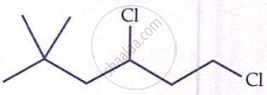 is ______.
is ______.
The IUPAC name of the compound shown below is: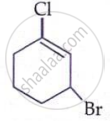
Ethylidene chloride is a/an ______.
