Advertisements
Advertisements
Question
When concentrated sulphuric acid was added to an unknown salt present in a test tube a brown gas (A) was evolved. This gas intensified when copper turnings were added to this test tube. On cooling, the gas (A) changed into a colourless solid (B).
1) Identify (A) and (B).
2) Write the structures of (A) and (B).
3) Why does gas (A) change to solid on cooling?
Advertisements
Solution
1) A = NO2 ; B = N2O4
2)
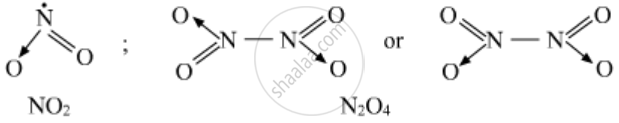
3) It converts into solid because of NO2 form dimer i.e N2O4 at low temperature.
APPEARS IN
RELATED QUESTIONS
Draw the structure of the following:
H2SO4
Why is `K_(a_2) "<<" K_(a_1)` for `H_2SO_4` in water?
Write chemical reactions for different steps in the manufacture of sulphuric acid by lead chamber process. Draw the structure of phosphorous pentachloride
Write a balanced chemical equation for the following reaction:
Sulphuric acid is treated with phosphorous.
Write structure and molecular formula for the following compounds:
a. Orthophosphoric acid
b. Sulphurous acid
The molecular formula of oleum is ____________.
Hot conc. \[\ce{H2SO4}\] acts as the moderately strong oxidising agent. It oxidises both metals and non-metals. Which of the following element is oxidised by conc. \[\ce{H2SO4}\] into two gaseous products?
Which of the following statements are correct?
(i) S – S bond is present in \[\ce{H2S2O6}\].
(ii) In peroxosulphuric acid \[\ce{(H2SO5)}\] sulphur is in +6 oxidation state.
(iii) Iron powder along with \[\ce{Al2O3}\] and \[\ce{K2O}\] is used as a catalyst in the preparation of \[\ce{NH3}\] by Haber’s process.
(iv) Change in enthalpy is positive for the preparation of \[\ce{SO3}\] by catalytic oxidation of \[\ce{SO2}\].
The number of dative bonds in sulphric acid molecule is ______.
What is the catalyst used for the oxidation of SO2 to SO3 in the lead chamber process for the manufacture of sulphuric acid?
