Advertisements
Advertisements
Question
What will be the order of reaction if the plot of log10[A] vs time is a straight line with a negative slope?
Numerical
Advertisements
Solution
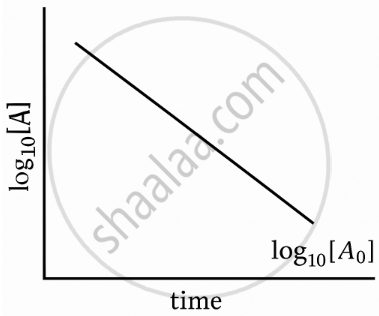
If the plot of log10[A] versus time (t) is a straight line with a negative slope, then the reaction is of first order.
For a first-order reaction, the integrated rate law is
`log_10[A] = log_10[A]_n - k/2.303 t`
This is a linear equation of the form
y = c − mt
Where:
y = log10[A],
t is time,
The slope `m = k/2.303` (positive constant),
So the slope of the graph is negative (because of the minus sign).
shaalaa.com
Is there an error in this question or solution?
