Advertisements
Advertisements
Questions
What is the shape of the curve obtained on plotting log10[A] against t for a first order reaction?
What is the graphical behaviour of a first order reaction on plotting log10[A] against t?
Advertisements
Solution
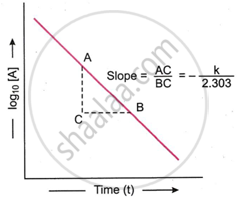
we know that
\[\ce{k = \frac{2.303}{t} log_10 \frac{[A]_0}{[A]}}\]
This equation can be written as
\[\ce{log_10[A] = - \frac{kt}{2.303} + log_10 [A]_0}\] ...(i)
The equation (i) is of the type y = mx + c and represents a straight line. Therefore, on plotting log10[A] against t, a straight line is obtained. The slope of the line is equal to \[\ce{\frac{- k}{2.303}}\] while the intercept of the line on log10[A] axis is equal to log10[A]0. Thus,
Slope of the line = \[\ce{\frac{AC}{BC} = - \frac{k}{2.303}}\]
Intercept on log10[A] axis = log10[A]0
Thus, the value of k can be obtained from the slope of the line.
