Advertisements
Advertisements
Question
What is the coordination number in a square close packed structure in two dimensions?
Options
2
3
4
6
Advertisements
Solution
4
Explanation:
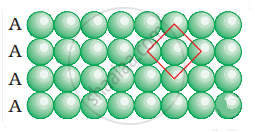
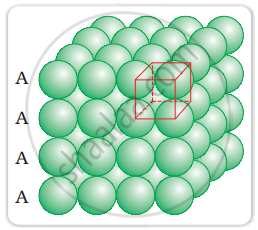
Coordination number in a square closed packed structure in two dimensions is equal to 4 is shown as:
APPEARS IN
RELATED QUESTIONS
A compound is formed by two elements M and N. The element N forms ccp and atoms of M occupy 1/3rdof tetrahedral voids. What is the formula of the compound?
A cubic solid is made of two elements P and Q. Atoms of Q are at the corners of the cube and P at the body-centre. What is the formula of the compound? What are the coordination numbers of P and Q?
Aluminium crystallises in a cubic close-packed structure. Its metallic radius is 125 pm.
- What is the length of the side of the unit cell?
- How many unit cells are there in 1.00 cm3 of aluminium?
Hexagonal close packed arrangement of ions is described as ____________.
The arrangement ABC ABC.... is referred to as ____________.
In NaCl structure ____________.
Match the type of packing given in Column I with the items given in Column II.
| Column I | Column II |
| (i) Square close packing in two dimensions |
(a) Triangular voids |
| (ii) Hexagonal close packing in two dimensions |
(b) Pattern of spheres is repeated in every fourth layer |
| (iii) Hexagonal close packing in three dimensions |
(c) Coordination number 4 |
| (iv) Cubic close packing in three dimensions |
(d) Pattern of sphere is repeated in alternate layers |
In the SF4 molecule, there are:
The octane number of Iso-octane is
The crystal system of a compound with unit cell dimensions a = 0.387, b = 0.387 and c = 0.504 nm and α = β = 90° and ϒ = 120° is
Total no. of voids in 0.5 mole of a compound forming hexagonal closed packed structure are:-
A compound forms hexagonal close-packed structure. What is the total number of voids in 0.5 mol of it? How many of these are tetrahedral voids?
Which of the following is frenkal effect.
The right option for the number of tetrahedral and octahedral voids in the hexagonal primitive unit cell is ______.
The empirical formula for a compound with a cubic close packed arrangement of anions and with cations occupying all the octahedral sites in AxB. The value of x is ______. (Integer answer)
The density of a pure substance 'X' whose atoms pack in cubic close pack arrangement is 1 g/cc. If all tetrahedral voids are occupied by 'Y' atoms. The value of '3a' is ______ g/cc, if the density of resulting solid is 'a' g/cc.
[Given: Atomic mass (X) = 30 g/mol, (Y) = 20 g/mol]
What fraction of one edge centred octahedral void lies in one unit cell of FCC?
