Advertisements
Advertisements
प्रश्न
What is the coordination number in a square close packed structure in two dimensions?
विकल्प
2
3
4
6
Advertisements
उत्तर
4
Explanation:
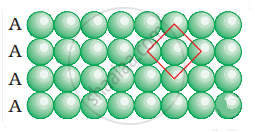
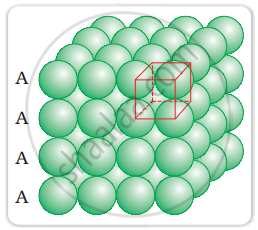
Coordination number in a square closed packed structure in two dimensions is equal to 4 is shown as:
APPEARS IN
संबंधित प्रश्न
What is the two dimensional coordination number of a molecule in square close packed layer?
A compound forms hexagonal close-packed structure. What is the total number of voids in 0.5 mol of it? How many of these are tetrahedral voids?
A cubic solid is made of two elements P and Q. Atoms of Q are at the corners of the cube and P at the body-centre. What is the formula of the compound? What are the coordination numbers of P and Q?
The arrangement ABC ABC.... is referred to as ____________.
In NaCl structure ____________.
How can you best describe the elongated octahedral structure of blue vitriol, CuSO4.5H2O?
In the SF4 molecule, there are:
The octane number of Iso-octane is
The packing efficiency of the two dimensional square unit cell shown below is:

A compound forms hexagonal close-packed structure. What is the total number of voids in 0.5 mol of it? How many of these are tetrahedral voids?
Co-ordination number of sodium ion Na+ in Nacl is:-
The number of tetrahedral and octahedral voids in a CCP array of 100 atoms are respectively:
The right option for the number of tetrahedral and octahedral voids in the hexagonal primitive unit cell is ______.
The right options for the number of tetrahedral and octahedral voids in the hexagonal primitive unit cells is ______.
Element 'B' forms ccp structures and A occupies half of the octahedral voids, while oxygen atoms occupy all the tetrahedral voids. The structure of bimetallic oxide is ______.
The density of a pure substance 'X' whose atoms pack in cubic close pack arrangement is 1 g/cc. If all tetrahedral voids are occupied by 'Y' atoms. The value of '3a' is ______ g/cc, if the density of resulting solid is 'a' g/cc.
[Given: Atomic mass (X) = 30 g/mol, (Y) = 20 g/mol]
What fraction of one edge centred octahedral void lies in one unit cell of FCC?
