Advertisements
Advertisements
Question
What is catenation? How does carbon form catenated compounds?
Advertisements
Solution
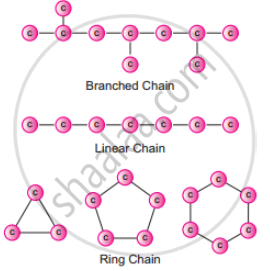
Catenation is the binding of an element to itself or with other elements through covalent bonds to form open chain or closed chain compounds. .
Carbon is the most common element which undergoes catenation and forms long chain compounds. Carbon atom links repeatedly to itself through covalent bond to form linear chain, branched chain or ring structure. This property of carbon itself is the reason for the presence of large number of organic carbon compounds. So organic chemistry essentially deals with catenated carbon compounds.
Catenation in carbon
APPEARS IN
RELATED QUESTIONS
The chemical properties of an organic compound are largely decided by the ______ and the physical properties of an organic compound are largely decided by the ______.
CHO is the functional group of an ______.
Name three compounds formed by ethene and give one use of each compound.
How will you identify?
A Soluble carbonate
Name the following:
A green coloured carbonate.
Give the structure of alcohol with 4 carbon atoms.
Write a short note on graphene.
Why do elements show allotropy?
Define:
Allotropy
Give the structure of alcohol with 4 carbon atoms.
