Advertisements
Advertisements
प्रश्न
What is catenation? How does carbon form catenated compounds?
Advertisements
उत्तर
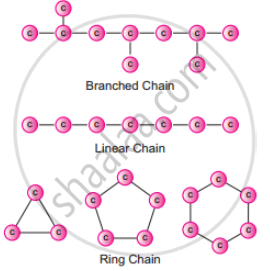
Catenation is the binding of an element to itself or with other elements through covalent bonds to form open chain or closed chain compounds. .
Carbon is the most common element which undergoes catenation and forms long chain compounds. Carbon atom links repeatedly to itself through covalent bond to form linear chain, branched chain or ring structure. This property of carbon itself is the reason for the presence of large number of organic carbon compounds. So organic chemistry essentially deals with catenated carbon compounds.
Catenation in carbon
APPEARS IN
संबंधित प्रश्न
The chemical properties of an organic compound are largely decided by the ______ and the physical properties of an organic compound are largely decided by the ______.
Give the reaction of chlorine with ethane and name the product formed.
What do you observe when ethene is passed through an alkaline KMnO4 solution?
The molecules of alkene family are represented by a general formula CnH2n.
Answer the following:
(i) What do n and 2n signify?
(ii) What is the name of alkene when n=4?
(iii) What is the molecular formula of the alkene, if there are six
hydrogen atoms in it?
(iv) Write the molecular and structural formula of the first member of
alkene family.
(v) Write the molecular formulae of lower and higher homologues of
an alkene which contains four carbon atoms.
How will you identify?
A Soluble carbonate
Name the following:
A green coloured carbonate.
What is tetravalency?
Why do elements show allotropy?
Define:
Allotrope
Give the structure of alcohol with 4 carbon atoms.
