Advertisements
Advertisements
Question
What do you observe when caustic soda solution is added to the following solution: first a little and then in excess : CuSO4
Advertisements
Solution
Addition of Caustic soda to CuSO4 Solution :
\[\ce{CuSO4 + 2NaOH ->\underset{\text{Pale blue ppt.}}{ Cu(OH)2 v + Na2SO4}}\]
APPEARS IN
RELATED QUESTIONS
A compound X (having the vinegar-like smell) when treated with ethanol in the presence of the acid Z, gives a compound Y which has a fruity smell.
The reaction is:
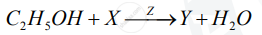
1) Identify Y and Z.
2) Write the structural formula of X.
3) Name the above reaction.
Name two colourless metal ions.
Name a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
Write balanced equation for a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
What do you observe when caustic soda solution is added to the following solution: first a little and then in excess : Pb(NO3)2
Name : A yellow monoxide that dissolves in hot and concentrated caustic alkali.
Fill in the blank.
To distinguish soluble salts of zinc and lead ______ [NaOH / NH4OH]can be used.
One chemical test that would enable you to distinguish between the following pair of chemicals. Describe what happens with each chemical or state 'no visible reaction'.
Calcium nitrate solution and zinc nitrate solution.
State your observation when zinc chloride solution, zinc nitrate solution, and zinc sulphate solutions are added individually to barium chloride solution.
Write balanced equations for a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
