Advertisements
Advertisements
Question
One chemical test that would enable you to distinguish between the following pair of chemicals. Describe what happens with each chemical or state 'no visible reaction'.
Calcium nitrate solution and zinc nitrate solution.
Advertisements
Solution
Calcium nitrate solution and zinc nitrate solution can be distinguished by sodium hydroxide. Sodium hydroxide reacts with zinc nitrate and zinc hydroxide is formed which is soluble in excess of sodium hydroxide.
\[\ce{Zn(NO3)2(aq) + 2NaOH(aq) -> Zn(OH)2(s) + 2NaNO3(aq)}\]
Sodium hydroxide reacts with calcium nitrate and forms white ppt. of calcium hydroxide insoluble in excess of sodium hydroxide.
\[\ce{Ca(NO3)2 + 2NaOH -> Ca(OH)2 + 2NaNO3}\]
APPEARS IN
RELATED QUESTIONS
Choose the most appropriate answer from the following list of oxides which fit the description
[SO2, SiO2, Al2O3, MgO, CO, Na2O]
An amphoteric oxide
A compound X (having the vinegar-like smell) when treated with ethanol in the presence of the acid Z, gives a compound Y which has a fruity smell.
The reaction is:
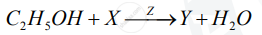
1) Identify Y and Z.
2) Write the structural formula of X.
3) Name the above reaction.
Give a chemical test to distinguish between the given pairs of chemicals:
Lead nitrate solution and Zinc nitrate solution
Name
a metallic hydroxide soluble in excess of NH4OH.
Name a metallic oxide soluble in excess of caustic soda solution.
Name a strong alkali.
Name two bases which are not alkalis but dissolves in strong alkalis.
Name the metal hydroxides which are:
Sparingly soluble
In caustic soda solution
What do you observe when caustic soda solution is added to the following solution: first a little and then in excess : CuSO4
Write balanced equations for a metal that evolves a gas which burns with a pop sound when boiled with alkali solutions.
