Advertisements
Advertisements
Question
What are the different types of thermodynamic systems?
Advertisements
Solution
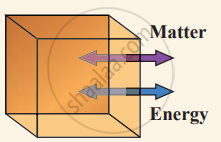
(i) Open system can exchange both matter and energy with the environment.
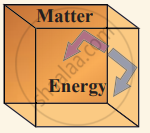
(ii) Closed system exchange energy but not matter with the environment.

(iii) Isolated system can exchange neither energy nor matter with the environment.
APPEARS IN
RELATED QUESTIONS
A hypothetical thermodynamic cycle is shown in the figure. Calculate the work done in 25 cycles.

The science of measuring temperatures is called?
A group of objects that can form a unit which may have the ability to exchange energy with its surrounding is called what?
On what basis a thermodynamic system can be classified?
Classify and explain the thermodynamic system.
What are intensive and extensive variables? Give examples.
Discuss the mechanical equilibrium.
Which of the following statements is correct for any thermodynamic system?
A gas in a closed container is heated with 10 J of energy, causing the lid of the container to rise 2 m with 3 N of force. What is the total change in energy of the system?
What are surroundings in thermodynamics?
