Advertisements
Advertisements
प्रश्न
What are the different types of thermodynamic systems?
Advertisements
उत्तर
(i) Open system can exchange both matter and energy with the environment.
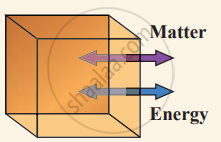
(ii) Closed system exchange energy but not matter with the environment.

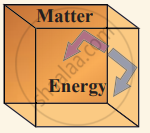
(iii) Isolated system can exchange neither energy nor matter with the environment.
APPEARS IN
संबंधित प्रश्न
A gas contained in a cylinder fitted with a frictionless piston expands against a constant external pressure of 1 atm from a volume of 5 liters to a volume of 10 liters. In doing so it absorbs 400J of thermal energy from its surroundings. Determine the change in the internal energy of the system.
A group of objects that can form a unit which may have the ability to exchange energy with its surrounding is called what?
On what basis a thermodynamic system can be classified?
An ideal gas passes from one equilibrium state (P1, V1, T1, N) to another equilibrium state (2P1, 3V1, T2, N). Then
A hot cup of coffee is kept on the table. After some time it attains a thermal equilibrium with the surroundings. By considering the air molecules in the room as a thermodynamic system, which of the following is true
Identify the state variables given here?
What is mean by state variable? Give example.
What is an equation of state? Give an example.
Discuss the chemical equilibrium.
An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume V1 and contains ideal gas at pressure P1 and temperature T1. The other chamber has volume V2 and contains ideal gas at pressure P2 and temperature T2. If the partition is removed without doing any work on the gases, calculate the final equilibrium temperature of the container.
