Advertisements
Advertisements
Question
What are n-type semiconductors? Why is the conductivity of doped n-type semiconductor higher than that of pure semiconductor? Explain with diagram.
Answer in Brief
Advertisements
Solution
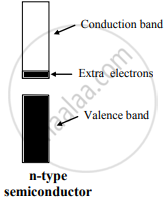
- An extrinsic semiconductor, which is obtained by adding group 15 element to an intrinsic semiconductor which belongs to group 14, is called an n-type semiconductor.
e.g. Silicon doped with phosphorus - n-type semiconductor contains an increased number of electrons in the conduction band.
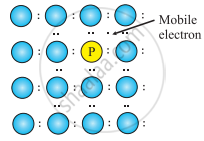
- Consider the doping of Si with phosphorus. Si has a crystal structure in which each Si atom is linked tetrahedrally to four other Si atoms. When a small quantity of phosphorous is added to pure Si, the P atoms occupy some vacant sites in the lattice in place of Si atoms. The overall crystal structure of Si remains unchanged.
Four of the five valence electrons of P are utilized in bonding the closest to four Si atoms. Thus, P has one extra electron than needed for bonding. Therefore, Si doped with P has more number of electrons in the conduction band than those in the conduction band in pure Si. Thus, the conductivity of Si-doped with P is higher than that of pure Si. The electrons in the conduction band move under the influence of an applied potential and conduct electricity.
P atom occupying regular site of Si atom
shaalaa.com
Is there an error in this question or solution?
