Advertisements
Advertisements
Questions
Explain with a diagram the Frenkel defect.
Describe the Frenkel defect found in ionic crystals.
Explain the following term with suitable examples:
Frenkel defect
Diagram
Explain
Very Long Answer
Advertisements
Solution
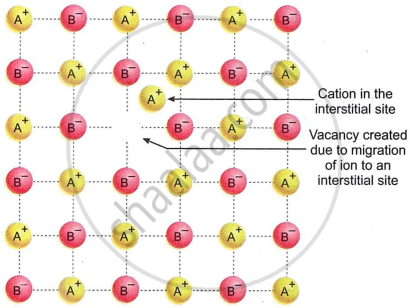
- A Frenkel defect happens when an ion in an ionic compound is absent from its usual spot in the lattice and instead sits in a space between the lattice points.
- The cations are usually smaller than anions. Therefore, the cations occupy interstitial sites. The smaller cation shifts from its usual location to the interstitial space.
- It, therefore, creates a vacancy defect at its original position and an interstitial defect at its new location in the same crystal.
- A Frenkel defect can be regarded as the combination of a vacancy defect and an interstitial defect.
- In alkali metal halides, Frenkel flaws don't happen very often because the alkali metal cations are too big to fit into smaller spaces between the atoms.
- Frenkel defects are common in silver halides, e.g., AgCl, AgBr, CaF2 and Agl. They are also found in ZnS crystal.
shaalaa.com
Is there an error in this question or solution?
