Advertisements
Advertisements
Question
Underline the incorrectly balanced compound equation and rewrite the correct equation.
\[\ce{FeCl3 + 3NH4OH->2NH4Cl + Fe(OH)3}\]
Advertisements
Solution
Correct equation is: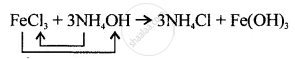
APPEARS IN
RELATED QUESTIONS
Balance the following equation:
N2 + H2 → NH3
Balance the following equation:
Na2O + H2O → NaOH
Balance the following equation:
\[\ce{K + H2O->KOH + H2}\]
Balance the following important equation:
Al + Cl2 → AlCl3
Balance the following important equation:
NH3 + O2 → NO + H2O (catalytic oxidation of NH3)
Which of the following is true for an unbalanced chemical equation?
Solid calcium oxide reacts vigorously with water to form calcium hydroxide accompanied by liberation of heat. This process is called slaking of lime. Calcium hydroxide dissolves in water to form a solution called lime water. Which among the following is (are) true about slaking of lime and the solution formed?
- It is an endothermic reaction
- It is an exothermic reaction
- The pH of the resulting solution will be more than seven
- The pH of the resulting solution will be less than seven
In which of the following chemical equations, the abbreviations represent the correct states of the reactants and products involved at reaction temperature?
Which of the following are combination reactions?
- `2"KClO"_3 overset("Heat")(->) 2"KCl" + 3"O"_2`
- `"MgO" + "H"_2"O" -> "Mg"("OH")_2`
- `4"Al" + 3"O"_2 -> 2"Al"_2"O"_3`
- `"Zn" + "FeSO"_4 -> "ZnSO"_4 + "Fe"`
Write a balanced chemical equation of the following reaction and also classify them.
Lead acetate solution is treated with dilute hydrochloric acid to form lead chloride and acetic acid solution.
