Advertisements
Advertisements
Question
Three solutions P, Q and R have pH value of 3.5, 5.2 and 12.2 respectively. Which one of these is a:
1) Weak acid?
2) Strong alkali?
Advertisements
Solution
1) Q having pH 5.2 is the weak acid
2) R having pH 12.2 is strong alkali.
APPEARS IN
RELATED QUESTIONS
What happens to the H+ ions, of the solution, when an acid is neutralised?
What happens when bases react with non-metal oxides? Explain with the help of an example. What does this reaction tell us about the nature of non-metal oxides?
Write the chemical formula of ammonium chloride. Explain why an aqueous solution of ammonium chloride is acidic in nature? Illustrate your answer with the help of a chemical equation.
Choosing the correct words given in brackets, complete the sentence given below:
An acid is a compound which, when dissolved in water gives ______ ( hydronium / hydroxide) ions as the only ______ (positive/negative) ions.
Which of the following is not a mineral acid?
State whether the following statement is true or false. Correct the false statement.
A compound if acidic will turn all indicators red.
Fill in the cross word given in figure with the help of the clues provided.
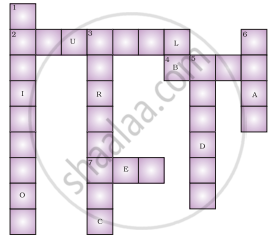
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour in basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Complete the following equation.
ZnO + 2HCl → ______ + ______.
Acidic or basic solutions are good conductors of electricity. Justify your answer.
Select an oxide which dissolves in water forming an acid.
