Advertisements
Advertisements
Question
The smoke from a fire looks white. Which of the following statements is true?
1) Molecules of the smoke are bigger than the wavelength of light
2) Molecules of the smoke are smaller than the wavelength of light.
Advertisements
Solution
The smoke from a fire looks white because molecules of the smoke are bigger than the wavelength of light, scatter the light off all wavelengths of white light to the same extent.
Therefore, statement-1 is true.
APPEARS IN
RELATED QUESTIONS
Which colour of white light is deviated by a glass prism
- the most and
- the least?
How does the speed of light in glass change on increasing the wavelength of light?
Calculate the frequency of yellow light of wavelength 550 nm. The speed of light is 3 × 108 m/s-1.
Fig shows a thin beam of white light from a source S striking on one face of a prism.

- Complete the diagram to show the effect of the prism on the beam and to show what is seen on the screen.
- If a slit is placed in between the prism and the screen to pass only the light of green colour, what will you then observe on the screen?
- What conclusion do you draw from the observation in part (b) above?
Write the seven prominent colours present in white light spectrum in order of their increasing wavelength.
Name the seven prominent colours of the white light spectrum in order of their increasing frequencies.
Name four colours of the spectrum of white light which have wavelength longer than blue light.
Which colour of white light is deviated at least?
An imaginary material reflects red light, scatters green light, transmits blue light, and absorbs yellow, orange, and violet light. A cube of this material is shown in the given diagram.
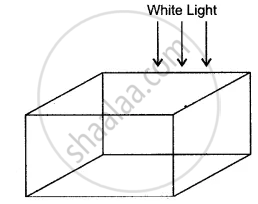
The surface is illuminated with a beam of white light. What colour will this cube appear?
(i) from above,
(ii) from below and
(iii) from the sides?
What is the range of wavelength of the spectrum of white light nm?
