Advertisements
Advertisements
Question
The ground state energy of a hydrogen atom is −13.6 eV. What are the kinetic and potential energies of the electron in this state?
Advertisements
Solution
Ground state energy of hydrogen atom, E = −13.6 eV
This is the total energy of a hydrogen atom. Kinetic energy is equal to the negative of the total energy.
Kinetic energy = − E
= − (−13.6)
= 13.6 eV
Potential energy is equal to the negative of two times the kinetic energy.
Potential energy = − 2 × Kinetic energy
= − 2 × (13.6)
= −27.2 eV
APPEARS IN
RELATED QUESTIONS
Given the ground state energy E0 = - 13.6 eV and Bohr radius a0 = 0.53 Å. Find out how the de Broglie wavelength associated with the electron orbiting in the ground state would change when it jumps into the first excited state.
A 12.5 eV electron beam is used to bombard gaseous hydrogen at room temperature. Upto which energy level the hydrogen atoms would be excited? Calculate the wavelengths of the first member of Lyman and first member of Balmer series.
A difference of 2.3 eV separates two energy levels in an atom. What is the frequency of radiation emitted when the atom makes a transition from the upper level to the lower level?
The total energy of an electron in the first excited state of the hydrogen atom is about −3.4 eV.
What is the kinetic energy of the electron in this state?
The total energy of an electron in the first excited state of the hydrogen atom is about −3.4 eV.
Which of the answers above would change if the choice of the zero of potential energy is changed?
Obtain the first Bohr’s radius and the ground state energy of a muonic hydrogen atom [i.e., an atom in which a negatively charged muon (μ−) of mass about 207 me orbits around a proton].
Wavelengths of the first lines of the Lyman series, Paschen series and Balmer series, in hydrogen spectrum are denoted by `lambda_L, lambda_P and lambda_B` respectively. Arrange these wavelengths in increasing order.
A 12.3 eV electron beam is used to bombard gaseous hydrogen at room temperature. Upto which energy level the hydrogen atoms would be excited?
Calculate the wavelengths of the second member of Lyman series and second member of Balmer series.
Calculate the minimum amount of energy which a gamma ray photon should have for the production of an electron and a positron pair..
The Ionisation energy of hydrogen atom is 3.6 ev The ionisation energy of helium atom would be
Energy of an electron at infinity from nucleus is ______.
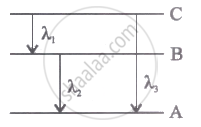
Energy levels A, B, C of acertain atom corresponding to increasing value of energy, i.e., EA< E8 < Ee. If λ1, λ2 and λ3 are the wavelength of radiations corresponding to the transitions C to B, B to A and C to A respectively, which of the following statements is correct?
Two H atoms in the ground state collide inelastically. The maximum amount by which their combined kinetic energy is reduced is ______.
Radiation coming from transitions n = 2 to n = 1 of hydrogen atoms fall on He+ ions in n = 1 and n = 2 states. The possible transition of helium ions as they absorb energy from the radiation is ______.
Which of the following is true for X-rays?
