Advertisements
Advertisements
Question
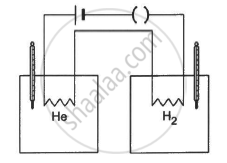
The figure shows two vessels with adiabatic walls, one containing 0.1 g of helium (γ = 1.67, M = 4 g mol−1) and the other containing some amount of hydrogen (γ = 1.4, M = 2 g mol−1). Initially, the temperatures of the two gases are equal. The gases are electrically heated for some time during which equal amounts of heat are given to the two gases. It is found that the temperatures rise through the same amount in the two vessels. Calculate the mass of hydrogen.
Advertisements
Solution
Given:
Mass of He, mHe = 0.1 g
γ1 = 1.67
Molecular weight of He, MHe = 4 g/mol
MH2 = ?
MH2 = 2 g/mol
γ2 = 1.4
Since it is an adiabatic environment and the system is not dong any external work, the amount of heat given will be used up entirely to raise its internal energy.
For He, dQ = dU = nCvdT ...(i)
`= "m"/4 xx "R"/(gamma-1) xx "d""T"`
`= 0.1 /4 xx "R"/((1.67 -1)) xx "d""T"`
For H2, dQ =dU = nCvdT ...(ii)
=`"m"/2 xx "R"/(gamma-1) xx "d""T"`
=`"m"/2 xx "R"/(1.4-1) xx "d""T"`
where m is the required mass of H2.
Since equal amount of heat is given to both gases; so dQ is same in both eq (i) and (ii), we get
`0.1/4 xx "R"/0.67""d""T"`
`= "m"/2 xx "R"/0.4 xx"d""T"`
`= m = 0.1/2 xx 0.4 /0.67`
`=> m =0.0298 ≈ 0.03 "g"`
APPEARS IN
RELATED QUESTIONS
A metre long narrow bore held horizontally (and closed at one end) contains a 76 cm long mercury thread, which traps a 15 cm column of air. What happens if the tube is held vertically with the open end at the bottom?
Given below are densities of some solids and liquids. Give rough estimates of the size of their atoms:
| Substance | Atomic Mass (u) | Density (103 Kg m-3) |
| Carbon (diamond) | 12.01 | 2.22 |
| Gold | 197.00 | 19.32 |
| Nitrogen (liquid) | 14.01 | 1.00 |
| Lithium | 6.94 | 0.53 |
| Fluorine (liquid) | 19.00 | 1.14 |
[Hint: Assume the atoms to be ‘tightly packed’ in a solid or liquid phase, and use the known value of Avogadro’s number. You should, however, not take the actual numbers you obtain for various atomic sizes too literally. Because of the crudeness of the tight packing approximation, the results only indicate that atomic sizes are in the range of a few Å].
Does a gas have just two specific heat capacities or more than two? Is the number of specific heat capacities of a gas countable?
Can we define specific heat capacity for an adiabatic process?
Does a solid also have two kinds of molar heat capacities Cp and Cv? If yes, is Cp > Cv? Or is Cp − Cv = R?
In an isothermal process on an ideal gas, the pressure increases by 0.5%. The volume decreases by about
Consider the processes A and B shown in the figure. It is possible that
Three identical adiabatic containers A, B and C contain helium, neon and oxygen, respectively, at equal pressure. The gases are pushed to half their original volumes.
(a) The final temperatures in the three containers will be the same.
(b) The final pressures in the three containers will be the same.
(c) The pressures of helium and neon will be the same but that of oxygen will be different.
(d) The temperatures of helium and neon will be the same but that of oxygen will be different.
5 g of a gas is contained in a rigid container and is heated from 15°C to 25°C. Specific heat capacity of the gas at constant volume is 0.172 cal g−1 °C−1 and the mechanical equivalent of heat is 4.2 J cal−1. Calculate the change in the internal energy of the gas
An ideal gas expands from 100 cm3 to 200 cm3 at a constant pressure of 2.0 × 105 Pa when 50 J of heat is supplied to it. Calculate (a) the change in internal energy of the gas (b) the number of moles in the gas if the initial temperature is 300 K (c) the molar heat capacity Cp at constant pressure and (d) the molar heat capacity Cv at constant volume.
4.0 g of helium occupies 22400 cm3 at STP. The specific heat capacity of helium at constant pressure is 5.0 cal K−1 mol−1. Calculate the speed of sound in helium at STP.
Standing waves of frequency 5.0 kHz are produced in a tube filled with oxygen at 300 K. The separation between the consecutive nodes is 3.3 cm. Calculate the specific heat capacities Cp and Cv of the gas.
An engine takes in 5 moles of air at 20°C and 1 atm, and compresses it adiabatically to `1/10^"th"` of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be X kJ. The value of X to the nearest integer is ______.
If at same temperature and pressure, the densities for two diatomic gases are respectively d1 and d2 then the ratio of velocities of sound in these gases will be ______.
