Advertisements
Advertisements
Question
The density of gold is 19 g/cm3. Find the volume of 95 g of gold.
Advertisements
Solution
ρ (density) = 19 g/cm3
V (volume) = ?
m (mass) = 95 g
`ρ = m/V`
`V = m/ρ`
= `95/19`
V = 5 cm3
Answer: Volume of gold = 5 cm3.
APPEARS IN
RELATED QUESTIONS
Define density. What is the SI unit of density ?
Define relative density. What is the SI unit of relative density ?
A piece of steel has a volume of 12 cm3, and a mass of 96 g. What is its density in g/cm3 ?
When a golf ball is lowered into a measuring cylinder containing water, the water level rises by 30 cm3 when the ball is completely submerged. If the mass of ball in air is 33 g, find its density.
A metal in which even iron can float is :
A solid of density 900 kg/m3 floats in oil as shown in the given diagram. The oil floats on water of density 1000 kg/m3 as shown. The density of oil kg/m3 could be :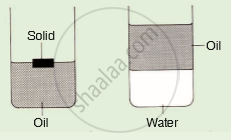
The density of water is 1000 kg/m3 and the density of copper is 8900 kg/m3. Which of the following statements is incorrect?
The diagrams represent four measuring cylinders containing liquids. The mass and volume of the liquid in each cylinder are stated. Which two measuring cylinder could contain an identical liquid?
To find out the relative density of the substance, with respect to the density of water ______C is taken.
The density Bottle is also called ______.
